ZetaView® Concentration Zeta Potential Fluorescence Size Subpopulations See what you measure
Viruses
Viruses are nanoscale pathogens containing nucleic acids that can infect all life forms such as humans, animals, bacteria, and plants. Bacteria infecting viruses are called bacteriophages or phages. Since viruses do not have their own metabolism, they depend on a host cell for reproduction. For this reason, they are not counted as living organisms.
Each virus can exist in two different forms, either inside its host cell or as independent particles called virions. Virions consist of genetic material containing information about their blueprint, and a surrounding protein envelope called a capsid. Depending on the type of nucleic acid they are made of, viruses are classified as DNA or RNA, single-stranded or double-stranded. Some virions also possess a lipid bilayer membrane called an envelope. The origin of this envelope is a membrane of its host cell. For example, the HI virus (the causative agent of AIDS) or SARS-CoV-2 are enveloped viruses.
The morphology of virions varies greatly. Their size varies between 15 – 30 nm for small viruses like Circoviridae and 400 – 500 nm for the large giant viruses. Because of their size range, they can be perfectly measured by NTA. Their shape is determined by their protein coat and can be spherical, but also helical, spherical or isometrical.
Viruses and diseases
Viruses are responsible for a high number of different diseases. In humans, some prominent examples are COVID-19, hepatitis, AIDS or Ebola.
Not every viral infection causes a disease. If the immune system is able to recognize and fight the pathogen due to vaccination or previous infection, a disease is avoided. However, because viruses lack an effective proof-reading strategy for their nucleic acid replication in the host cell, they have a high mutation rate. The resulting strains of a virus are often not recognized by the immune cells and can therefore cause a disease. This high mutation rate leads to a finely tuned adaptation to the host and can cause a jump to another species, such as from animals to humans, known as zoonosis. For example, the HI virus was first discovered in monkeys and most likely adapted to humans as hosts by eating infected meat.
The better the adaptation of the virus to its host, the lower the mortality, since the virus is dependent on its host. Therefore, coexistence of the virus with its host can be achieved, often with little or no symptoms. A typical example of a well-adapted virus is the herpes simplex virus, which has an infection rate of 65 – 90 % in adults worldwide and cannot be cleared by the immune system after a first infection. Herpes simplex infection shows mild symptoms from time to time when the immune system is weakened, but can cause severe symptoms in immunocompromised persons.
Viruses are also responsible for about 17% of all human cancer cases. The so-called oncoviruses responsible for this lead to uncontrolled cellular growth by altering intracellular signalling pathways, which causes cancer. A well-known example is the hepatitis B virus causing hepatocarcinoma or the human papillomavirus, which causes cervical cancer, for example.
Virologists are working to understand the host-virus-relationship and unravel the mechanisms of the immune response to a viral attack. Therefore, they need efficient methods to quantify the viral load, or titer required to cause an infection. One common method is the so-called plaque assay, in which different dilutions of virions are added to a monolayer of cultured cells. Due to the cytopathic effect of the viral infection, the cells become lysed, which is visible as colourless holes after a staining the cell monolayer. As a result, the number of plaque forming units (PFU) can be determined and is expressed as PFU/ml.
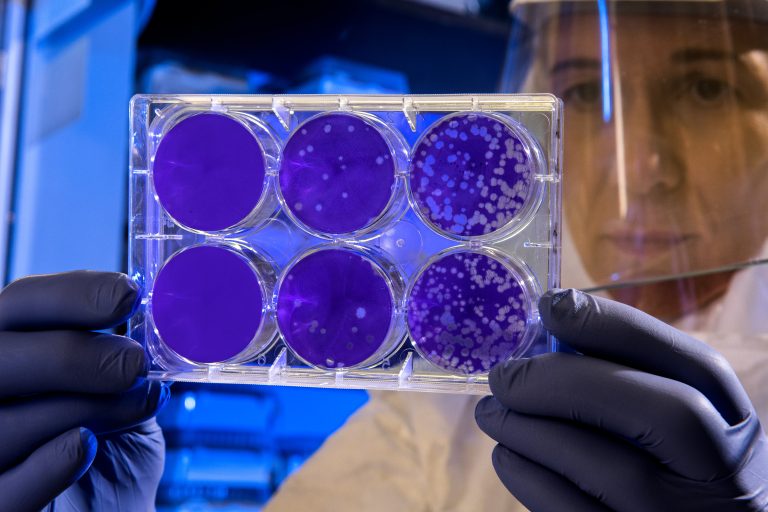
As an alternative test, the Median Tissue Culture Infectious Dose assay (TCID50) can be performed. In this assay, several varying concentrations of virions are added to a cell monolayer. After a defined time, the plates are examined for cytopathic effects of cell death. The concentration that results in 50% cytopathic effects is defined as TCID50 and is expressed as TCID50/ml.
You can read a comparison of both methods here.
Both methods lack the ability to quantify the total concentration of virions as they only indicate infections, but not particles. Therefore, the Nanoparticle Tracking Analysis technique is an important addition to the established methods. Virions can be quantified within a few minutes. An advantage of the Nanoparticle Tracking Analysis technique is that it can also measure the non-infectious fraction, which gives a more reliable result for the replication rate of a virus.
By using fluorescent Nanoparticle Tracking Analysis (F-NTA), defined virions can be stained and thus identified from complex samples such as patient material. Alternatively, F-NTA can be used to analyse the amount of successfully loaded (nucleic acid-containing) virions.
Since particle size can be measured with Nanoparticle Tracking Analysis, viral aggregation, which alters viral behaviour, can also be visualized.
Viruses as therapeutic approach
Viral Vector Vaccines
In addition to their pathogenic effect, viruses are also used for therapeutic purposes. For example, viruses can be used as vehicles for transporting genetic material into target cells. These viral vectors have been known to the public since the Corona pandemic, when viral vector vaccines directed against SARS-CoV-2 were invented. They transport the DNA or RNA of a pathogen into the target cell, where a protein is expressed which is then recognized by the immune system. Thereby the immune system is trained and able to prevent an infection by this pathogen afterwards.
Gene Therapy
Beyond their use as vaccines, viral vectors can also be used for gene therapy. This involves curing hereditary diseases by replacing the dysfunctional gene with a working one. However, since these approaches show undesirable side effects, their use has so far been limited to research and clinical trials.
Oncolytic Cancer Therapy
Viruses are known to cause cancer, but they can also cure cancer. These healing viruses are called oncolytic viruses and have diverse mechanisms to fight tumor cells. Some oncolytic viruses kill tumor cells directly by lysis. Others support the immune system to react against tumor cells, which results in suppressed tumour growth or even tumor shrinkage. Or they induce tumor suppressor genes in cancer cells, which reduces or stops tumor growth. This so-called oncolytic cancer therapy is the subject of numerous clinical studies.
Phage Therapy
The oldest medical use of viruses is phage therapy, in which bacteriophages are used to cure bacterial infections. It was invented as early as 1915 by Frederick Twort. After the invention of antibiotics, phage therapy became less interesting although it has some interesting advantages. It is very specific and therefore has fewer side effects compared to antibiotics. However, the interest in phage therapy is rising due to the increase in antibiotic resistance.
Nanoparticle Tracking Analysis is a common technique to ensure the quality of production of all these viral therapeutics.
Application Notes
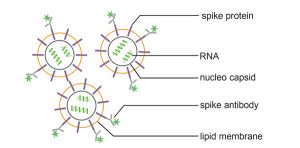
Specific detection of SARS-CoV-2 corona virus from cell culture supernatants by fluorescence-based Nanoparticle Tracking Analysis
In this new application note we describe a quick determination of the physical titer of coronaviruses by fluorescence-based Nanoparticle Tracking Analysis and specific spike antibody recognition.
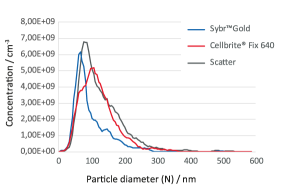
Rapid determination of purity, integrity and titer
of viruses by Fluorescence Nanoparticle Tracking
Analysis (F-NTA) using the bacteriophage Phi6
Determination of the titer of viruses and bacteriophages is an indispensable key technology in virological research and for diagnostic purposes. Depending on the method used, the measurements are either qualitative or quantitative, very time-consuming, and do not provide information about integrity or aggregation behaviour of the virus particles. Nanoparticle Tracking Analysis with the Particle Metrix ZetaView® instrument allows the user to perform a rapid concentration determination of virus particles. Using the bacteriophage Phi6 as an example, we show how titer, purity and integrity of phage particles can be measured quickly and reliably using the fluorescence detection capability of the Particle Metrix ZetaView® instrument.
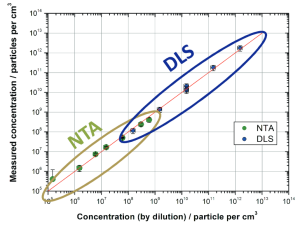
Three in One: Concentration, Size & Zeta Potential measured on sub-visible protein suspensions
The preparation of high-value biological material, such as proteins, requires a multi-step procedure of separation and purification. Each step must be optimized to reach full efficiency of yield.
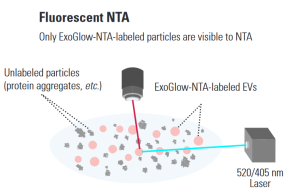
Detect only extracellular vesicles with Nanoparticle Tracking Analysis (NTA)
Making a great exosome research tool even better, SBI has developed ExoGlow TM -NTA, a proprietary dye that enables fluorescent analysis of only the extracellular vesicles (EVs) present in a heterogeneous sample.
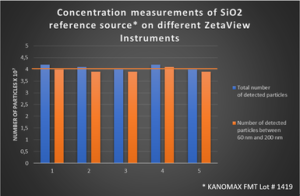
Reliability of concentration measurements with the Particle Metrix ZetaView® Nanoparticle Tracking Analyzer
Concentration levels of extracellular vesicles have a high clinical potential as a reference for disease.
Phenotyping of EVs by Multi-Wavelength Fluorescence Nanoparticle Tracking Analysis
F-NTA of fluorescent labelled bionanoparticles is an efficient technique for quantification of size and total concentration.
Publications
A Mildly Acidic Environment Alters Pseudomonas aeruginosa Virulence and Causes Remodeling of the Bacterial Surface
Mozaheb, Negar; Rasouli, Paria; Kaur, Mandeep; van der Smissen, Patrick; Larrouy-Maumus, Gerald; Mingeot-Leclercq
Microbiology Spectrum
2023
ApplicationsConcentration MeasurementExtracellular VesiclesMicrovesiclesPathogeneSize MeasurementVesicles
acidic pHenvelopelow pHmembrane vesiclesPseudomonas aeruginosa
Full TextA simple and available measurement of oncosEV dsDNA to protein ratio as a potential tumor marker
Sun, Yifan; Li, Miao; Zhang, Xiaoshan; Xu, Dongjie; Wu, Jie; Gu, Xinrui; Khan, Adeel; Shen, Han; Li, Zhiyang
BMC Cancer
2023
ApplicationsBiomarkersCancerConcentration MeasurementDiagnosticsExtracellular VesiclesSize MeasurementSmall Extracellular VesiclesVesicles
Double-stranded DNANucleic acid to protein ratioSmall extracellular vesiclestumor diagnosis
Full TextAnnexin A5 Inhibits Endothelial Inflammation Induced by Lipopolysaccharide-Activated Platelets and Microvesicles via Phosphatidylserine Binding
Tschirhart, Brent J.; Lu, Xiangru; Gomes, Janice; Chandrabalan, Arundhasa; Bell, Gillian; Hess, David A.; Xing, Guangxin; Ling, Hong; Burger, Dylan; Feng, Qingping
Pharmaceuticals
2023
ApplicationsConcentration MeasurementExtracellular VesiclesMicrovesiclesSize MeasurementVesicles
annexin A5endothelial cellsendotoxemiaextracellular vesiclesinflammationplateletssepsis
Full TextBacillus cereus extracellular vesicles act as shuttles for biologically active multicomponent enterotoxins
Buchacher, Tanja; Digruber, Astrid; Kanzler, Markus; Del Favero, Giorgia; Ehling-Schulz, Monika
Cell communication and signaling : CCS 21 (1)
2023
ApplicationsConcentration MeasurementDrug DeliveryExosomesExtracellular VesiclesSize MeasurementVesicles
3D-SIM microscopyBacillus cereusextracellular vesiclesHost-pathogen interactionMulticomponent toxinNon-hemolytic enterotoxinSMase
Full TextBiodegradable Mg alloy modified with bioactive exosomes for cardiovascular stent application
Hou, Ya-chen; Li, Jing-an; Cao, Chang; Su, Chang; Qin, Zhen; Zhang, Ge; Guo, Jia-cheng; Tang, Jun-nan; Zhang, Jin-ying; Guan, Shao-kang
Journal of Magnesium and Alloys
2023
ApplicationsCardiovascular DiseasesConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Full TextBiophysical and proteomic analyses of Pseudomonas syringae pv. tomato DC3000 extracellular vesicles suggest adaptive functions during plant infection
Janda, Martin; Rybak, Katarzyna; Krassini, Laura; Meng, Chen; Feitosa-Junior, Oséias; Stigliano, Egidio; Szulc, Beata; Sklenar, Jan; Menke, Frank L. H.; Malone, Jacob G.; Brachmann, Andreas; Klingl, Andreas; Ludwig, Christina; Robatzek, Silke
mBio
2023
ApplicationsConcentration MeasurementExtracellular VesiclesPathogeneSize MeasurementVesiclesZeta Potential
Arabidopsis thalianaEVsextracellular vesiclesnanoparticle tracking analysisNTApattern-triggered immunityproteomicsPTIPto DC3000
Full TextBioprinted constructs that simulate nerve–bone crosstalk to improve microenvironment for bone repair
Wang, Tianchang; Li, Wentao; Zhang, Yuxin; Xu, Xiang; Qiang, Lei; Miao, Weiqiang; Yue, Xiaokun; Jiao, Xin; Zhou, Xianhao; Ma, Zhenjiang; Li, Shuai; Ding, Muliang; Zhu, Junfeng; Yang, Chi; Wang, Hui; Li, Tao; Sun, Xin; Wang, Jinwu
Bioactive Materials
2023
ApplicationsExtracellular VesiclesMicrovesiclesSize MeasurementVesicles
BioprintingexosomesMicroenvironmentNerve–bone crosstalkSchwann cells
Full TextBovine milk-derived extracellular vesicles enhance doxorubicin sensitivity in triple negative breast cancer cells by targeting metabolism and STAT signalling
Sanwlani, Rahul; Kang, Taeyoung; Gummadi, Sriram; Nedeva, Christina; Ang, Ching-Seng; Mathivanan, Suresh
Proteomics
2023
ApplicationsBreast CancerCancerConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
bovine milk-derived extracellular vesiclescancer chemotherapydietary extracellular vesiclesspecies crosstalkstat signallingtriple-negative breast cancer
Full TextChange in cytokine profiles released by mast cells mediated by lung cancer-derived exosome activation may contribute to cancer-associated coagulation disorders
Ben, Suqin; Huang, Xiulin; Shi, Yongxin; Xu, Ziheng; Xiao, Hui
Cell communication and signaling : CCS 21 (1)
2023
ApplicationsCancerExosomesExtracellular VesiclesLung CancerSize MeasurementVesicles
cancer associated coagulation disorderscancer-associated thrombosisexosomeslung cancerMast cells
Full TextChanges in urinary exosomal protein CALM1 may serve as an early noninvasive biomarker for diagnosing diabetic kidney disease
Li, Tao; Ci Liu, Tian; Liu, Na; Zhang, Man
Clinica Chimica Acta; international journal of clinical chemistry
2023
ApplicationsBiomarkersConcentration MeasurementDiabetes TherapyExosomesExtracellular VesiclesSize MeasurementVesicles
BiomarkerCalcium signal pathwayCALM1Diabetic kidney diseaseurinary exosomes
Full TextCharacteristics of size-exclusion chromatography enriched porcine follicular fluid extracellular vesicles
Kamińska, Kinga; Godakumara, Kasun; Świderska, Bianka; Malinowska, Agata; Midekessa, Getnet; Sofińska, Kamila; Barbasz, Jakub; Fazeli, Alireza; Grzesiak, Malgorzata
Theriogenology
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesiclesZeta Potential
Atomic Force Microscopyextracellular vesiclesNanoparticle Tracking Analysis (NTA)OvaryPigSize-exclusion chromatography
Full TextCharacterization of cannabis strain‑plant‑derived extracellular vesicles as potential biomarkers
Ipinmoroti, Ayodeji O.; Turner, Ja'kayla; Bellenger, Elizabeth J.; Crenshaw, Brennetta J.; Xu, Junhuan; Reeves, Caitlin; Ajayi, Olufemi; Li, Ting; Matthews, Qiana
Protoplasma
2023
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
CannabidiolCannabisCultivarFiberPlant-derived extracellular vesicles
Full TextCord Blood Plasma and Placental Mesenchymal Stem Cells-Derived Exosomes Increase Ex Vivo Expansion of Human Cord Blood Hematopoietic Stem Cells While Maintaining Their Stemness
Teleb, Rasha S.; Abdul-Hafez, Amal; Othman, Amira; Ahmed, Ahmed El-Abd; Elsaid, Abdelrahman A.; Arif, Hattan; Zarea, Ahmed A.; Abdulmageed, Mohammed; Mohamed, Hend; Ibrahim, Sherif Abdelfattah; Thiruvenkataramani, Ranga P.; Mohamed, Tarek; Kanada, Masamitsu; Madhukar, Burra V.; Arellano, Myrna Gonzalez; Sayed, Mohammed M.; Qubaisy, Heba M.; Omar, Said A.
Cells
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
exosomesextracellular vesicles (EVs)hematopoietic stem cellsmesenchymal stem cellsplacentaumbilical cordumbilical cord blood transplantation
Full TextDepletion of abundant plasma proteins for extracellular vesicle proteome characterization: benefits and pitfalls
Reymond, Sandrine; Gruaz, Lyssia; Sanchez, Jean-Charles
Analytical and Bioanalytical Chemistry
2023
ApplicationsBiomarkersConcentration MeasurementDiagnosticsExosomesExtracellular VesiclesSize MeasurementVesicles
extracellular vesiclesPlasma depletionproteomicsSize-exclusion chromatography
Full TextDifferent Biofluids, Small Extracellular Vesicles or Exosomes: Structural Analysis in Atherosclerotic Cardiovascular Disease Using Electron Microscopy Techniques
Sharma, Pratibha; Dhamija, Rajinder K.; Nag, Tapas Chandra; Roy, Ambuj; Inampudi, Krishna Kishore
Microscopy and Microanalysis
2023
ApplicationsExosomesExtracellular VesiclesSmall Extracellular VesiclesVesicles
atherosclerosiselectron microscopyexosomenanoparticle tracking analysisPlasmasalivaSmall extracellular vesiclesurine
Full TextEffects of lysate/tissue storage at -80°C on subsequently extracted EVs of epithelial ovarian cancer tissue origins
Shen, Shizhen; Shen, Zhangjin; Wang, Conghui; Wu, Xiaodong; Wang, Lingfang; Ye, Lele; Zhang, Songfa; Cheng, Xiaodong
iScience
2023
ApplicationsCancerConcentration MeasurementExtracellular VesiclesOvarian CancerVesicles
epithelial ovarian cancerextracellular vesicleslysate -80°C storagetissue -80°C storage
Full TextEffects of Plasma-Derived Exosomal miRNA-19b-3p on Treg/T Helper 17 Cell Imbalance in Behçet’s Uveitis
Jiang, Qingyan; Wang, Qingfeng; Tan, Shiyao; Cai, Jinyu; Ye, Xingsheng; Su, Guannan; Yang, Peizeng
Investigative ophthalmology & visual science
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Behçet’s uveitisCD46miR-19b-3pplasma exosomesTreg/Th17 cell imbalance
Full TextEmploying nanoparticle tracking analysis of salivary neuronal exosomes for early detection of neurodegenerative diseases
Sharma, Vaibhav; Nikolajeff, Fredrik; Kumar, Saroj
Translational Neurodegeneration
2023
Alzheimer’s diseaseApplicationsExosomesExtracellular VesiclesParkinson's DiseaseVesicles
Alzheimer’s Diseasediagnosisexosomesextracellular vesiclesnanoparticle tracking analysisNeurodegenerative diseaseParkinson's diseasePrognosis
Full TextEndoplasmin Is a Hypoxia-Inducible Endoplasmic Reticulum-Derived Cargo of Extracellular Vesicles Released by Cardiac Cell Lines
Koncz, Anna; Turiák, Lilla; Németh, Krisztina; Lenzinger, Dorina; Bárkai, Tünde; Lőrincz, Péter; Zelenyánszki, Helga; Vukman, Krisztina V.; Buzás, Edit I.; Visnovitz, Tamás
Membranes 13 (4)
2023
ApplicationsConcentration MeasurementDiagnosticsExosomesSize MeasurementSmall Extracellular VesiclesVesicles
cardiomyocyteEndoplasmic reticulum stressexosomeextracellular vesiclemedium EVsmall EV
Full TextEquine mesenchymal stem cell derived extracellular vesicle immunopathology biomarker discovery
Reynolds, David E.; Vallapureddy, Phoebe; Morales, Renee‐Tyler T.; Oh, Daniel; Pan, Menghan; Chintapula, Uday; Linardi, Renata L.; Gaesser, Angela M.; Ortved, Kyla; Ko, Jina
Journal of Extracellular Biology
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
biomarker discoveryequineextracellular vesicleimmunomodulationmesenchymal stem celltherapeutic
Full TextExosomal miRNA-223-3p derived from tumor associated macrophages promotes pulmonary metastasis of breast cancer 4T1 cells
Wang, Ziyuan; Zhang, Chen; Guo, Jian; Wang, Wei; Si, Qin; Chen, Chong; Luo, Yunping; Duan, Zhaojun
Translational Oncology
2023
ApplicationsBreast CancerCancerConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Cbx5exosomesmetastasismiR-223-3pTAMs
Full TextExosomes Derived from Dermatophagoides farinae Induce Allergic Airway Inflammation
Yang, Ting; Xu, Zuyu; Yu, Jinyan; Liu, Jiaxi; Wang, Wei; Hong, Shanchao
Microbiology Spectrum
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesPathogeneSize MeasurementVesicles
allergic airway inflammationalveolar macrophagebronchial epithelial cellcomparative transcriptomicsDermatophagoides farinaeexosome
Full TextExtracellular Vesicle ITGAM and ITGB2 Mediate Severe Acute Pancreatitis-Related Acute Lung Injury
Hu, Qian; Zhang, Shu; Yang, Yue; Li, Juan; Kang, Hongxin; Tang, Wenfu; Lyon, Christopher J.; Wan, Meihua
ACS nano
2023
ApplicationsExosomesExtracellular VesiclesSize MeasurementVesicles
acute lung injuryextracellular vesiclesintegrin α Mintegrin β 2pulmonary microvascular endothelial cellssevere acute pancreatitis
Full TextExtracellular vesicles derived from mouse adipose-derived mesenchymal stem cells promote diabetic corneal epithelial wound healing through NGF/TrkA pathway activation involving dendritic cells
Wang, Guifang; Zeng, Li; Gong, Can; Gong, Xileyuan; Zhu, Tupeng; Zhu, Yujie
Experimental Eye Research
2023
ApplicationsDiabetes TherapyExosomesExtracellular VesiclesSize MeasurementVesicles
Dendritic cellsDiabetic keratopathymADSC-EVsNGF/TrkA pathway
Full TextHuman adipose tissue‑derived small extracellular vesicles promote soft tissue repair through modulating M1‑to‑M2 polarization of macrophages
Dong, Jia; Wu, Bin; Tian, Weidong
Stem cell research & therapy 14 (1)
2023
ApplicationsExtracellular VesiclesSize MeasurementSmall Extracellular VesiclesVesicles
Adipose tissue regenerationHuman adipose tissue-derived small extracellular vesicleMacrophage polarizationSkin wound healing
Full TextHyperthermia promotes M1 polarization of macrophages via exosome‑mediated HSPB8 transfer in triple negative breast cancer
Di Xu; Liu, Zhen; Liang, Ming-Xing; Chen, Wen-Quan; Fei, Yin-Jiao; Yang, Su-Jin; Wu, Yang; Zhang, Wei; Tang, Jin-Hai
Discover. Oncology 14 (1)
2023
ApplicationsBreast CancerCancerConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
exosomesHeat shock protein family B (small) member 8 (HSPB8)hyperthermiaM1 macrophages polarizationtriple-negative breast cancer
Full TextIdentification and analysis of the DNA content of small extracellular vesicles isolated from Leishmania parasites
Ibarra-Meneses, Ana Victoria; Amin, Atia; Dong, George; Olivier, Martin; Langlais, David; Fernandez-Prada, Christopher
STAR protocols
2023
ApplicationsConcentration MeasurementExtracellular VesiclesIsolation TechniquesParasitesSize MeasurementSmall Extracellular VesiclesVesicles
extracellular vesiclesLeishmania parasitesnanoparticle tracking analysispurificationultracentrifugation
Full TextImproved Wound Healing and Skin Regeneration Ability of 3,2′-Dihydroxyflavone-Treated Mesenchymal Stem Cell-Derived Extracellular Vesicles
Kim, Sehee; Shin, Yeokyung; Choi, Yujin; Lim, Kyung-Min; Jeong, Yeojin; Dayem, Ahmed Abdal; Lee, Yoonjoo; An, Jongyub; Song, Kwonwoo; Jang, Soo Bin; Cho, Ssang-Goo
IJMS (International Journal of Molecular Sciences) 24
2023
ApplicationsExosomesExtracellular VesiclesSize MeasurementVesicles
anti-inflammationEVsflavonoidMEK/ERK signalMSCswound healing
Full TextIn situ formed scaffold with royal jelly-derived extracellular vesicles for wound healing
Tan, Dehong; Zhu, Wenxiang; Liu, Lujie; Pan, Yuanwei; Xu, Yangtao; Huang, Qinqin; Li, Lingling; Rao, Lang
Theranostics
2023
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesWound healing
Biomaterialsextracellular vesiclesHydrogelRoyal Jellywound healing
Full TextIndependent human mesenchymal stromal cellderived extracellular vesicle preparations differentially attenuate symptoms in an advanced murine graft-versus-host disease model
Madel, Rabea J.; Börger, Verena; Dittrich, Robin; Bremer, Michel; Tertel, Tobias; Phuong, Nhi Ngo Thi; Baba, Hideo A.; Kordelas, Lambros; Staubach, Simon; Stein, Frank; Haberkant, Per; Hackl, Matthias; Grillari, Regina; Grillari, Johannes; Buer, Jan; Horn, Peter A.; Westendorf, Astrid M.; Brandau, Sven; Kirschning, Carsten J.; Giebel, Bernd
Cytotherapy
2023
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
exosomesextracellular vesiclesgraft-versus-host-diseaseheterogeneitymesenchymal stem cellsmesenchymal stromal cells
Full TextM2 Macrophages-Derived Exosomal miRNA-23a-3p Promotes the Progression of Oral Squamous Cell Carcinoma by Targeting PTEN
Li, Jun; Bao, Yongjie; Peng, Sisi; Jiang, Chao; Zhu, Luying; Zou, Sihai; Xu, Jie; Li, Yong
CIMB (Current Issues in Molecular Biology)
2023
ApplicationsCancerConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
exosomeM2 macrophagesmacrophagesMiR-23a-3pOSCCPTEN
Full TextMesenchymal stromal cell exosomes enhance dental pulp cell functions and promote pulp-dentin regeneration
Shi, Jiajun; Teo, Kristeen Ye Wen; Zhang, Shipin; Lai, Ruenn Chai; Rosa, Vinicius; Tong, Huei Jinn; Duggal, Mandeep S.; Lim, Sai Kiang; Toh, Wei Seong
Biomaterials and Biosystems
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Dental pulpexosomesextracellular vesiclesMesenchymal stromal/stem cellsRegeneration
Full TextNovel neutrophil extracellular trap-related mechanisms in diabetic wounds inspire a promising treatment strategy with hypoxia-challenged small extracellular vesicles
Chu, Ziqiang; Huang, Qilin; Ma, Kui; Liu, Xi; Zhang, Wenhua; Cui, Shengnan; Wei, Qian; Gao, Huanhuan; Hu, Wenzhi; Wang, Zihao; Meng, Sheng; Tian, Lige; Li, Haihong; Fu, Xiaobing; Zhang, Cuiping
Bioactive materials 27
2023
ApplicationsConcentration MeasurementDiabetes TherapyExtracellular VesiclesSize MeasurementSmall Extracellular VesiclesVesicles
Diabetic wound healingEndoplasmic reticulum stresshypoxiaNeutrophil extracellular trapsSmall extracellular vesicles
Full TextOviduct epithelial cell‑derived extracellular vesicles promote the developmental competence of IVF porcine embryos
Fang, Xun; Bang, Seonggyu; Tanga, Bereket Molla; Seo, Chaerim; Zhou, Dongjie; Seong, Gyeonghwan; Saadeldin, Islam M.; Lee, Sanghoon; Cui, Xiang-Shun; Cho, Jongki
Molecular medicine reports 27
2023
ApplicationsConcentration MeasurementExtracellular VesiclesFertilizationSize MeasurementVesicles
embryoepithelial cellsextracellular vesiclesin vitro fertilizationoviductporcine
Full TextPhosphatidylserine-positive extracellular vesicles boost effector CD8+ T cell responses during viral infection
Rausch, Lisa; Flaskamp, Lavinia; Ashokkumar, Ashretha; Trefzer, Anne; Ried, Christine; Buchholz, Veit R.; Obst, Reinhard; Straub, Tobias; Brocker, Thomas; Kranich, Jan
Proceedings of the National Academy of Sciences
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
CD8 T cellsexosomesextracellular vesiclesLCMVPhosphatidylserine
Full TextPlasma-derived extracellular vesicles released after endurance exercise exert cardioprotective activity through the activation of antioxidant pathways
Lisi, Veronica; Senesi, Giorgia; Bertola, Nadia; Pecoraro, Matteo; Bolis, Sara; Gualerzi, Alice; Picciolini, Silvia; Raimondi, Andrea; Fantini, Cristina; Moretti, Elisa; Parisi, Attilio; Sgrò, Paolo; Di Luigi, Luigi; Geiger, Roger; Ravera, Silvia; Vassalli, Giuseppe; Caporossi, Daniela; Balbi, Carolina
Redox Biology
2023
ApplicationsCardiovascular DiseasesConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementSmall Extracellular VesiclesVesicles
Antioxidant activitycardioprotectionCatalytic activityextracellular vesiclesPhysical exerciseRedox capacity
Full TextPlasma‑derived small extracellular vesicles unleash the angiogenic potential in head and neck cancer patients
Tengler, Luisa; Schütz, Julia; Tiedtke, Moritz; Jablonska, Jadwiga; Theodoraki, Marie-Nicole; Nitschke, Katja; Weiß, Christel; Seiz, Elena; Affolter, Annette; Jungbauer, Frederic; Lammert, Anne; Rotter, Nicole; Ludwig, Sonja
Molecular Medicine
2023
ApplicationsCancerConcentration MeasurementExtracellular VesiclesSize MeasurementSmall Extracellular VesiclesVesicles
Angiogenesisendothelial cellsexosomesHead and neck cancerPlasmaSmall extracellular vesicles
Full TextProtein Biomarker Discovery Studies on Urinary sEV Fractions Separated with UF-SEC for the First Diagnosis and Detection of Recurrence in Bladder Cancer Patients
Jordaens, Stephanie; Oeyen, Eline; Willems, Hanny; Ameye, Filip; Wachter, Stefan de; Pauwels, Patrick; Mertens, Inge
Biomolecules
2023
ApplicationsBiomarkersCancerConcentration MeasurementExtracellular VesiclesSize MeasurementSmall Extracellular VesiclesVesicles
Biomarkerextracellular vesiclesLiquid biopsynon-muscle-invasive bladder cancer (NMIBC)oncologyproteomicsurine
Full TextQuantitative imaging of vesicle-protein interactions reveals close cooperation among proteins
Cha, Minkwon; Jeong, Sang Hyeok; Jung, Jaehun; Baeg, Yoonjin; Park, Sung-Soo; Bae, Seoyoon; Lim, Chan Seok; Park, Jun Hyuk; Lee, Jie-Oh; Gho, Yong Song; Oh, Seung Wook; Shon, Min Ju
J. Extracell. Vesicles (Journal of Extracellular Vesicles) Volume 12, Issue 5
2023
ApplicationsExtracellular VesiclesSize MeasurementVesicles
CD9cell-derived vesiclesICAM-1integrinvesicle-protein interactions
Full TextRegulation of Hsa-miR-4639-5p expression and its potential role in the pathogenesis of Parkinson’s disease
He, Lu; Chen, Yimeng; Lin, Suzhen; Shen, Ruinan; Pan, Hong; Zhou, Yifan; Wang, Ying; Chen, Shengdi; Ding, Jianqing
Aging cell
2023
ApplicationsExosomesExtracellular VesiclesParkinson's DiseaseSize MeasurementVesicles
DJ-1exosomehistone acetylationhsa-miR-4639-5pParkinson's diseasepromoter
Full TextSCARA5 in bone marrow stromal cell-derived exosomes inhibits colorectal cancer progression by inactivating the PI3K/Akt pathway
Fang, Yu; Wu, Feng; Shang, Guoyin; Yin, Changqing
Genomics
2023
ApplicationsCancerColorectal CancerExosomesExtracellular VesiclesSize MeasurementVesicles
Bone marrow stromal cellcolorectal cancerexosomesPI3K/AktSCARA5
Full TextSelective pharmacological inhibition alters human carcinoma lung cell-derived extracellular vesicle formation
Ipinmoroti, Ayodeji O.; Pandit, Rachana; Crenshaw, Brennetta J.; Sims, Brian; Matthews, Qiana L.
Heliyon 9
2023
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementSmall Extracellular VesiclesVesicles
ESCRTextracellular vesiclesInhibitionPathway
Full TextSequencing and bioinformatics analysis of miRNA from rat endplate chondrogenic exosomes
Chen, Xue-Wu; Li, Qiu-Wei; Wang, Hong
Exp Ther Med (Experimental and Therapeutic Medicine) 25 (6)
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesmiRNASize MeasurementVesicles
epigeneticexosomeintervertebral disc degenerationmiRNA
Full TextSUR7 deletion in Candida albicans impacts extracellular vesicle features and delivery of virulence factors
McKenna, James A.; Garcia‐Ceron, Donovan; Bleackley, Mark R.; Yu, Long; Bulone, Vincent; Anderson, Marilyn
Journal of Extracellular Biology
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesFungiSize MeasurementVesicles
Candida albicansextracellular vesiclesSur7virulence
Full TextVariability of extracellular vesicle release during storage of red blood cell concentrates is associated with differential membrane alterations, including loss of cholesterol-enriched domains
Ghodsi, Marine; Cloos, Anne-Sophie; Mozaheb, Negar; van der Smissen, Patrick; Henriet, Patrick; Pierreux, Christophe E.; Cellier, Nicolas; Mingeot, Marie-Paule; Najdovski, Tomé; Tyteca, Donatienne
Frontiers in physiology
2023
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesVesicles
cholesterolintracellular ATPmembrane microviscosityoxidative stressphosphatidylserine surface exposurered blood cell transfusionspectrin networksphingomyelin-enriched domains
Full TextAdipose-derived mesenchymal stem cell-loaded β-chitin nanofiber hydrogel promote wound healing in rats
Liu, Y., Liu, Y., Wu, M. et al.
Journal of Materials Science: Materials in Medicine
2022
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
stem cellswound healingβ-chitin nanofibers
Full TextBlocking exosomal secretion aggravates 1,4-Benzoquinone-induced mitochondrial fission activated by the AMPK/MFF/Drp1 pathway in HL-60 cells
Lu, Fangfang; Zhang, Qianqian; Zhang, Mengyan; Sun, Shuqiang; Yang, Xinjun; Yan, Hongtao
Journal of applied toxicology
2022
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
AMP-activatedexosomemitochondriamitochondrial dynamicsprotein kinase
Full TextConfocal microscopy analysis reveals that only a small proportion of extracellular vesicles are successfully labelled with commonly utilised staining methods
Melling, G.E., Conlon, R., Pantazi, P. et al.
Sci Rep 12
2022
ApplicationsBiomarkersExosomesExtracellular VesiclesFluorescenceSize MeasurementVesicles
C5-maleimide-Alexa633PKH26protocol optimization
Full TextExosomal lncRNA ATB Derived from Ovarian Cancer Cells Promotes Angiogenesis via Regulating miR-204-3p/TGFβR2 Axis
Yuan D, Guo T, Zhu D, Ge H, Zhao Y, Huang A, Wang X, Cao X, He C, Qian H, Yu H
Cancer Management and Research
2022
ApplicationsCancerExosomesExtracellular VesiclesOvarian CancerSize MeasurementVesicles
Full TextExosomal miR-17-3p Alleviates Programmed Necrosis in Cardiac Ischemia/Reperfusion Injury by Regulating TIMP3 Expression
Zhuyuan Liu, Didi Zhu, Fuchao Yu, Mingming Yang, Dan Huang, Zhenjun Ji, Wenbin Lu, and Genshan Ma
Oxidative Medicine and Cellular Longevity: Oxidative Stress and Programmed Cell Death in Cardiovascular Diseases
2022
ApplicationsCardiovascular DiseasesConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
miRNAmiaMyocardial ischemia/reperfusion injuryReperfusio
Full TextExtracellular Vesicles Derived from SIPA1high Breast Cancer Cells Enhance Macrophage Infiltration and Cancer Metastasis through Myosin-9
Feng, Lingyun; Weng, Jun; Yao, Chenguang; Wang, Ruyuan; Wang, Ning; Zhang, Yilei; Tanaka, Yoshimasa; Su, Li
Biology
2022
ApplicationsExtracellular VesiclesSize MeasurementVesicles
Breast Cancerextracellular vesiclesmacrophagemyosin-9SIPA1
Full TextFetal Membranes Contribute to Drug Transport across the Feto-Maternal Interface Utilizing the Breast Cancer Resistance Protein (BCRP)
Ananthkumar Kammala, Meagan Benson, Esha Ganguly, Enkhtuya Radnaa, Talar Kechichian, Lauren Richardson and Ramkumar Menon
life: New Insights into the Placental and Placental Membrane Pathophysiology of Preterm Birth
2022
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
breast cancer resistance proteindrug transportationpregnancy
Full TextHuman Bone Marrow Stromal Cell Exosomes Ameliorate Periodontitis
Yue, C.; Cao, J.; Wong, A.; Kim, J. H.; Alam, S.; Luong, G.; Talegaonkar, S.; Schwartz, Z.; Boyan, B. D.; Giannobile, W. V.; Sahingur, S. E.; Lin, Z.
Journal of dental research
2022
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
cell therapyexosomesexperimental periodontitishuman bone marrow stromal cellsmiRNAperiodontal inflammation
Full TextNeutral Sphingomyelinase 2 Mediates Oxidative Stress Effects on Astrocyte Senescence and Synaptic Plasticity Transcripts
Zhu, Zhihui; Quadri, Zainuddin; Crivelli, Simone M.; Elsherbini, Ahmed; Zhang, Liping; Tripathi, Priyanka; Qin, Haiyan; Roush, Emily; Spassieva, Stefka D.; Nikolova-Karakashian, Mariana; McClintock, Timothy S.; Bieberich, Erhard
Molecular neurobiology
2022
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesVesicles
ceramideextracellular vesiclesGrin2boxidative stress
Full TextPlasma extracellular vesicle derived protein profile predicting and monitoring immunotherapeutic outcomes of gastric cancer
Zhang, Cheng; Chong, Xiaoyi; Jiang, Fangli; Gao, Jing; Chen, Yang; Jia, Keren; Fan, Meng; Liu, Xuan; An, Jin; Li, Jian; Zhang, Xiaotian; Shen, Lin
Journal of Extracellular Vesicles
2022
ApplicationsExtracellular VesiclesSize MeasurementVesicles
extracellular vesicle derived protein profilegastric cancerimmunotherapyplasma based liquid biopsytherapeutic biomarker
Full TextProfiling Blood Serum Extracellular Vesicles in Plaque Psoriasis and Psoriatic Arthritis Patients Reveals Potential Disease Biomarkers
Lättekivi, Freddy; Guljavina, Irina; Midekessa, Getnet; Viil, Janeli; Heath, Paul R.; Bæk
International journal of molecular sciences
2022
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
Biomarkercirculating EVsmiRNApsoriatic arthritissurface proteome
Full TextProteomic and Biochemical Analysis of Extracellular Vesicles Isolated from Blood Serum of Patients with Melanoma
Kurg, Kristiina; Planken, Anu; Kurg, Reet
Separations
2022
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
extracellular vesiclesmelanomaPEG percipitationpurificationultracentrifugation
Full TextSecretory Autophagosomes from Alveolar Macrophages Exacerbate Acute Respiratory Distress Syndrome by Releasing IL-1β
Xu X, Liu X, Dong X, Qiu H, Yang Y, Liu L
Journal of Inflammation Research
2022
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
acute respiratory distress syndrome secretory autophagosomesalveolar macrophages
Full TextSerum-derived exosomes from SD rats induce inflammation in macrophages through the mTOR pathway
Wang, Ye; Liu, Yan-Ping; Sun, Zhan-Bing; Deng, Wei-Hua; Yuan, Xiao-Yan; Lei, Yuan-di; Cai, Ying; Huang, Lian; Zhang, Zhao-Hui
Journal of applied toxicology
2022
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Beryllium sulfateexosomesinflammationlung injurymTOR pathway
Full TextSmall RNA cargo armed extracellular vesicles alleviate periodontitis in OVX mice via M2 macrophage polarization
Danfeng Li, Yuyao Liu, Xinwei Lyu, Cheng Hu, Tong Yan, Jiayin Yan, Yuwei Liao, Xiaodan Chen, Jiali Tan
Chemical Engineering Journal
2022
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
Full TextSynergy of Human Platelet-Derived Extracellular Vesicles with Secretome Proteins Promotes Regenerative Functions
Fausto Gueths Gomes, André Cronemberger Andrade, Martin Wolf, Sarah Hochmann, Linda Krisch, Nicole Maeding, Christof Regl, Rodolphe Poupardin, Patricia Ebner-Peking, Christian G. Huber, Nicole Meisner-Kober, Katharina Schallmoser and Dirk Strunk
Biomedicines
2022
Extracellular VesiclesSize MeasurementVesiclesZeta Potential
EV coronahuman platelet lysateproteomics
Full TextAn engineered CD81-based combinatorial library for selecting recombinant binders to cell surface proteins: Laminin binding CD81 enhances cellular uptake of extracellular vesicles
Stefan Vogt, Madhusudhan Reddy Bobbili, Gerhard Stadlmayr, Katharina Stadlbauer, Jørgen Kjems, Florian Rüker, Johannes Grillari, Gordana Wozniak-Knopp
Journal of Extracellular Vesicles
2021
ApplicationsConcentration MeasurementDrug DeliveryExtracellular VesiclesSize MeasurementVesicles
Antibody StainingAntigen-binding CD81cellular uptake of EVsdrug deliveryev characterizationEV isolationEV uptakeHeLa derived EVsnanoparticle fluorescence stainingNTArecombinant EVsstem cell therapytargeted EVs
Full TextCharacterization of Extracellular Vesicles Labelled with a Lipophilic Dye Using Fluorescence Nanoparticle Tracking Analysis
Getnet Midekessa, Kasun Godakumara, Keerthie Dissanayake, Mohammad Mehedi Hasan, Qurat Ul Ain Reshi, Toonika Rinken, Alireza Fazeli
Membranes
2021
Extracellular VesiclesF-NTAFluorescenceVesicles
Biological Nanoparticlesev characterizationev measurementEV purificationf-NTAfluorescence NTAfluorescent labelinglipophilic dyesmembrane labelingnanoparticle charaterizationnanoparticle fluorescencenanoparticle labellingnanoparticle measurementzeta potential
Full TextDevelopment of surface engineered antigenic exosomes as vaccines for respiratory syncytial virus
Suyeon Hong, Shaobo Ruan, Zachary Greenberg, Mei He, and Jodi L. McGill
Scientific Reports
2021
ExosomesExtracellular VesiclesVaccinesVesiclesVirusesZeta Potential
dendritic cell exoxomesMHCRespiratory syncytial virus
Full TextEribulin and Paclitaxel Differentially Alter Extracellular Vesicles and Their Cargo from Triple-Negative Breast Cancer Cells
Petra J. Pederson, Huiyun Liang, Daria Filonov, Susan L. Mooberry
Cancers
2021
ApplicationsBreast CancerCancerConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
eribulinexosomesextracellular vesiclesmicrotubule targeting agentsmicrovesicles
Full TextFormation of a protein corona on the surface of extracellular vesicles in blood plasma
Eszter Á. Tóth, Lilla Turiák, Tamás Visnovitz, Csaba Cserép, Anett Mázló, Barbara W. Sódar, András I. Försönits, Gábor Petövári, Anna Sebestyén, Zsolt Komlósi, László Drahos, Ágnes Kittel, György Nagy, Attila Bácsi, Ádám Dénes, Yong Song Gho, Katalin É. Szabó-Taylor, Edit I. Buzás
Journal of Extracellular Vesicles
2021
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
EV isolationEV purificationEV size distributionEVs in blood plasmaEVs in rheumatoid arthritisexternal plasma protein cargomedium sized EVsmEVsNTAplasma protein contaminationplasma protein-coated EVsprotein coronaTHP1 derived EVs
Full TextHypoxia-stimulated ATM activation regulates autophagy-associated exosome release from cancer-associated fibroblasts to promote cancer cell invasion
Lei Xi, Meixi Peng, Shuiqing Liu, Yongcan Liu, Xueying Wan, Yixuan Hou, Yilu Qin, Liping Yang, Shanchun Chen, Huan Zeng, Yong Teng, Xiaojiang Cui, Manran Liu
Journal of Extracellular Vesicles
2021
ApplicationsBreast CancerCancerConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Autophagy-associated Exosome ReleaseBreast CancerCAF derived exosomescancer associated fibroblastsfibroblast derived exosomestumor developmenttumor progression
Full TextMicroRNA Candidate Biomarkers for Parkinson’s Disease and Idiopathic REM Sleep Behavior Disorder
Ligang Wu, Jun Liu, Yuanyuan Li, Ying Cao, Wei Liu, Hongdao Zhang, Aonan Zhao, Ningdi Luo
PREPRINT (Version 1) available at Research Square
2021
BiomarkersConcentration MeasurementExtracellular VesiclesParkinson's DiseaseSize MeasurementVesicles
EVsmall-seqidiopathic rapid eye movement sleep behavior disorder (iRBD)miRNAsncRNA
Full TextMicroRNAs are minor constituents of extracellular vesicles that are rarely delivered to target cells.
Albanese M, Chen Y-FA, Hüls C, Gärtner K, Tagawa T, Mejias-Perez E, et al.
PLOS GENETICS
2021
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
cargoCD63cell-cell communicationdonor cellexosomesextracellular vesiclesmiRNAPKH26 membrane dyetarget cellsviral miRNAs
Full TextP2RX7 inhibition reduces breast cancer induced osteolytic lesions – implications for bone metastasis
Karan M. Shah, Luke Tattersall, Aleana Hussain, Sarah C. Macfarlane, Alexander Williamson, Adelina E. Acosta-Martin, Janine T. Erler, Penelope D. Ottewell, Alison Gartland
bioRxiv
2021
ApplicationsBreast CancerCancerConcentration MeasurementExtracellular VesiclesMetastasisSize MeasurementVesicles
Full TextPorphyromonas gingivalis Provokes Exosome Secretion and Paracrine Immune Senescence in Bystander Dendritic Cells
Elsayed R, Elashiry M, Liu Y, El-Awady A, Hamrick M, Cutler CW
Front Cell Infect Microbiol.
2021
ApplicationsExosomesExtracellular VesiclesSize MeasurementVesicles
Dendritic cellsPeriodontitisPorphyromonas gingivalis
Full TextProteomic analysis of extracellular vesicles from tick hemolymph and uptake of extracellular vesicles by salivary glands and ovary cells
Xu, Zhengmao; Wang, Yanan; Sun, Meng; Zhou, Yongzhi; Cao, Jie; Zhang, Houshuang; Xuan, Xuenan; Zhou, Jinlin
Parasites & Vectors
2021
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
extracellular vesiclesFerritin-2HemolymphHyalomma asiaticumproteomicsRhipicephalus haemaphysaloide
Full TextTim4-Functionalized HBEV-Chip by Isolating Plasma-Derived Phosphatidylserine-Positive Small Extracellular Vesicles for Pan-Cancer Screening
Liu Yang, Xuedong Tong, Yong Zhang, Yan Li, Jiangwei Liu, Ruiling Yin, Dong Zeng, Yue Yuan, and Kun Deng
Advanced Materials Technology
2021
BiomarkersCancerConcentration MeasurementExtracellular VesiclesSize MeasurementSmall Extracellular VesiclesVesicles
pan-cancer screeningPhosphatidylserine
Full TextTumour-derived small extracellular vesicles suppress CD8+ T cell immune function by inhibiting SLC6A8-mediated creatine import in NPM1-mutated acute myeloid leukaemia
Meixi Peng, Jun Ren, Yipei Jing, Xueke Jiang, Qiaoling Xiao, Junpeng Huang, Yonghong Tao, Li Lei, Xin Wang, Zailin Yang, Zesong Yang, Qian Zhan, Can Lin, Guoxiang Jin, Xian Zhang, Ling Zhang
Journal of Extracellular Vesicles
2021
ApplicationsBreast CancerCancerConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
CD63paclitaxeltetraspanintriple-negative breast cancervinorelbine
Full TextExosomal miRNA-19b-3p of tubular epithelial cells promotes M1 macrophage activation in kidney injury.
Lin-Li Lv, Ye Feng, Min Wu, Bin Wang, Zuo-Lin Li, Xin Zhong, Wei-Jun Wu et al.
Cell Death & Differentiation
2020
BiomarkersCancerExosomesExtracellular VesiclesGastro-Esophageal AdenocarcinomaVesicles
Acute Kidney Injurychronic kidney injurychronic proteinuric kidney diseaseeGPC3exosoml miRNAsGlypican 3GPC3insterstitial inflammationkidney disease therapymicroRNAmiRNAsrenal exosomesTECTEC derived exosomestubular epithelial cellstubulointerstitial inflammation
Full TextExtracellular Vesicle-Contained microRNA of C. elegans as a Tool to Decipher the Molecular Basis of Nematode Parasitism
Thomas B. Duguet, Julien Soichot, Rostyslav Kuzyakiv, Lars Malmström and Lucienne Tritten
Frontiers in Cellular and Infection Microbiology
2020
Extracellular VesiclesmiRNAParasitesVesicles
Caenorhabditis elegansNematode Parasitism
Full TextSalivary extracellular vesicles inhibit Zika virus but not SARS-CoV-2 infection
Carina Conzelmann, Rüdiger Groß, Min Zou, Franziska Krüger, André Görgens, Manuela O Gustafsson, Samir El Andaloussi, Jan Münch & Janis A. Müller
Journal of Extracellular Vesicles
2020
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesViruses
Corona virusEV isolationEV purificationsaEVssalivary EVsSARS-CoV-2Zika virus
Full TextZeta Potential of Extracellular Vesicles: Toward Understanding the Attributes that Determine Colloidal Stability
Getnet Midekessa, Kasun Godakumara, James Ord, Janeli Viil, Freddy Lättekivi, Keerthie Dissanayake, Sergei Kopanchuk, Ago Rinken, Aneta Andronowska, Sourav Bhattacharjee, Toonika Rinken, and Alireza Fazeli
ACS Omega
2020
Extracellular VesiclesVesiclesZeta Potential
Buffer Influence on Zeta Potentialcolloidal stabilityJAr cell derived EVspH influence on zeta potentialsurface chargezeta potential
Full TextA dual signal amplification method for exosome detection based on DNA dendrimer self-assembly.
Meiling Gao, Fang He, Bin-Cheng Yin and Bang-Ce Ye
Analyst
2019
ApplicationsCancerConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
cancer biomarkerscancer diagnosiscancer metastasiscancer prognosiscancer therapycancer treatmentCD63CD63 aptamerdendrimerexosome detectionmagnetic beadstumor biomarkerstumor derived exosomestumor development
Full TextA PDGFRβ-based score predicts significant liver fibrosis in patients with chronic alcohol abuse, NAFLD and viral liver disease
Joeri Lambrecht, Stefaan Verhulst, Inge Mannaerts, Jan-Peter Sowa, Jan Best, Ali Canbay, Hendrik Reynaert, Leo A. van Grunsven
eBioMedicine
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesZeta Potential
diagnosisextracellular vesiclehepatic stellate cellliver disease diagnosisliver diseasesliver fibrosisplatelet derived growth factor receptor
Full TextAdipose tissue macrophage-derived exosomal miR-29a regulates obesity-associated insulin resistances
Ting Liu, Yu-Chen Sun, Peng Cheng, Hui-Ge Shao
Biochemical and Biophysical Research Communications
2019
ApplicationsConcentration MeasurementDiabetes TherapyExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Adipose Tissue MacrophagesATMsexosomesinsulin sensitivitymiR-29amiRNAsobesity-associated type 2 diabetesobesity-associatedi nsulin resistancePPAR-dtype 2 diabetes
Full TextAgeing characteristics of bone indicated by transcriptomic and exosomal proteomic analysis of cortical bone cells
Chenyang Zhang, Shuyu Xu, Shufan Zhang, Mengmeng Liu, Haiming Du, Ruinan Sun, Bo Jing and Yao Sun
Journal of Orthopaedic Surgery and Research
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Bone AgeingBone Degenerationexosomesgene expression profilingOLCSosteocyctesosteocytic lacunar-canalicular system
Full TextAptamer-based fluorescence polarization assay for separation-free exosome quantification
Zhen Zhang, Chuanhao Tang, Libo Zhao, Li Xu, Wei Zhou, Zaizai Dong, Yuqing Yang, Qiqi Xiec and Xiaohong Fang
Nanoscale
2019
ApplicationsCancerConcentration MeasurementExtracellular VesiclesTechnical NanoparticlesVesicles
cancer biomarkerscancer diagnosiscancer treatmentexosome identificationexosome quantificationexosomes in human plasmanon-invasive cancer diagnosisplasma derived exosomestumor derived exosomes
Full TextBMSCs protect against renal ischemia-reperfusion injury by secreting exosomes loaded with miR-199a-5p that target BIP to inhibit endoplasmic reticulum stress at the very early reperfusion stages
Chenyang Wang, Gongmin Zhu, Weiyang He, Hubin Yin, Fan Lin, Xin Gou and Xinyuan Li
The FASEB Journal
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
EVsextracellular vesiclesgum inflammationgum inflammation therapyM1 polarization of macrophagesmacrophagePDLSCperiodonitis therapyperiodontal diseaseperiodontal ligament stem cellperiodontitspolarization
Full TextBronchial epithelial cell extracellular vesicles ameliorate epithelial–mesenchymal transition in COPD pathogenesis by alleviating M2 macrophage polarization
Shengyang He MD, PhD, Duanni Chen MD, Mengyun Hu BS, Li Zhang MD, PhD, Caihong Liu MD et al.
Nanomedicine: Nanotechnology, Biology and Medicine
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Chronic lung diseaseCOPDepithelial-mesenchymal transitionexosomeextracellular vesiclesM2 macrophagesmiR-21small airway remodeling
Full TextCalcifying nanoparticles induce cytotoxicity mediated by ROS-JNK signaling pathways
Wu J, Tao Z, Deng Y, Liu Q1, Liu Y, Guan X, Wang X.
Urolithiasis
2019
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
ApoptosisAutophagyBcl-2 ExpressionCalcifying nanoparticles; Cytotoxicity; HK-2 cells; Kidney stone; ROSJNK-signalling pathwaymitochondrial membrane potential
Full TextChanges of microRNAs expression profiles from red swamp crayfish (Procambarus clarkia) hemolymph exosomes in response to WSSV infection
HuiYang, XixiLi, Jiaojun Ji, ChunyouYuan, Xiaojian Gao, Yingying Zhang, Cheng Lu, Fenggang Li, Xiaojun Zhang
Fish & Shellfish Immunology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesViruses
deep sequencingexosomeexosomesfishimmunologymicroRNAmiRNAP. clarkiaprocambarus clarkiashellfishWSSVWSSV virus
Full TextCharacterization and Antioxidant Activity of Short Linear Glucan–Lysine Nanoparticles Prepared by Maillard Reaction
Qianzhu Lin, Man Li, Liu Xiong. Lizhong Qiu, Xiliang Bian, Chunrui Sun, Qingjie Sun
Food Hydrocolloids
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Antioxidative Nanoparticlesdebranched starchdeliverydrug deliveryfree radical scavengeLS-NPslysinemaillard reactionmelanoidinsnutraceutical deliveryshort linear glucanSLGSLG-Lys nanoparticlesstability
Full TextChimeric peptide engineered exosomes for dual-stage light guided plasma membrane and nucleus targeted photodynamic therapy
Cadi Davies, Aidan J. Taylor, Abdi Elmi, Jody Winter, Janie Liaw, Anna D. Grabowska, Ozan Gundogdu, Brendan W. Wren, David J. Kelly and Nick Dorrell
Frontiers in Cellular and Infection Microbiology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesOuter Membrane VesiclesSize MeasurementVesicles
Dual-targetingexosomesLysosomal escapePhotochemical internalizationPhotodynamic therapy
Full TextDetection of AML-specific mutations in pediatric patient plasma using extracellular vesicle–derived RNA
Kunz, F., Kontopoulou, E., Reinhardt, K. et al.
Annals of Hematology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesZeta Potential
Acute Myeloid Leukemiachildhood acute myeloid leukemiaextracellular vesiclesleukemia biomarkerspediatric AMLRNA
Full TextEfficient isolation, biophysical characterisation and molecular composition of extracellular vesicles secreted by primary and immortalised cells of reproductive origin
Jamaludin NA, Thurston LM, Witek KJ, Meikle A, Basatvat S, Elliott S, Hunt S, Andronowska A, Fazeli A.
Theriogenology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesZeta Potential
establishment of pregnancyEVs intercelllar communicationimmortalised cellsmicroRNAmiRNAoviductal epithelialprimary cellstheriogenology
Full TextErythrocyte Membrane-camouflaged IR780 and DTX co-loading Polymeric Nanoparticles for Imaging-guided Cancer Photo-Chemo Combination Therapy
Qian Yang, Yao Xiao, Yanlong Yin, Gaoyin Li, Jinrong Peng
Molecular Pharmaceutics
2019
ApplicationsBreast CancerCancerConcentration MeasurementDrug DeliveryExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Breast Cancer Therapycancer therapycombination therapydrug deliverydual drug-loaded nanoparticlesdual model imaginglong circulationnanoparticle drug-loadingphoto-chemo combination therapytumor phototherapytumor-targeting drug delivery
Full TextExosomal miR-1229 derived from colorectal cancer cells promotes angiogenesis by targeting HIPK2
Hui-Ying Hu, Chen-Huan Yu, Huan-Huan Zhang, Song-Zhao Zhang, Wen-Ying Yu. Yang, Yang, Qin Chen
International Journal of Biological Macromolecules
2019
ApplicationsBiomarkersCancerColorectal CancerConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Angiogenesiscancer biomarkerscolorectal cancerCRCCRC angiogenesisCRC-derived exosomesdiagnostic biomarkerexomiRexomiR-1229exosomal biomarkersexosomal miRNATumor microenvironmentVEGFVEGF pathway
Full TextExosomal miR-499a-5p promotes cell proliferation, migration and EMT via mTOR signaling pathway in lung adenocarcinoma
Lambros Kordelas, Esther Schwich, Robin Dittrich, Peter A. Horn, Dietrich W. Beelen, Shan He, Ziming Li, Yongfeng Yu, Qingyu Zeng, Yirui Cheng, Wenxiang Ji, Weiliang Xia, Shun Lu
Experimental Cell Research
2019
ApplicationsCancerConcentration MeasurementExtracellular VesiclesLung CancerSize MeasurementTechnical NanoparticlesVesicles
cancer biomarkerscancer diagnosiscancer metastasiscarcinogenesisexosomal miRNAexosomeslung cancermicroRNAmiR499amiRNAmTOR pathwaytumor derived exosomes
Full TextExosome secreted from adipose-derived stem cells attenuates diabetic nephropathy by promoting autophagy flux and inhibiting apoptosis in podocytes
Juan Jin, Yifen Shi, Jianguang Gong, Li Zhao, Yiwen Li, Qiang He and He Huang
Stem Cell Research & Therapy
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Adipose-derived Stem CellsAutophagyAutophagy-associated Exosome Releasecell autophagydipose-derived stem cellsexosomesmiR-486podocyte damageSmad1/mTOR signaling
Full TextExosome-mediated targeted delivery of miR-210 for angiogenic therapy after cerebral ischemia in mice
Huixin Zhang, Jin Wu, Jiahuan Wu, Qi Fan, Jingchao Zhou, Junwen Wu, Sichen Liu, Jie Zang, Jinhai Ye, Ming Xiao, Tian Tian and Jun Gao
Journal of Nanobiotechnology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
AngiogenesisBrain Tissue Repaircerebral ischemiadrug deliveryendogenous delivery systemexosomesischemiaischemic strokemicroRNAmiR-210miRNAmiRNA deliverytargeted deliverytargeted EVs
Full TextExosomes derived from human umbilical cord mesenchymal stromal cells deliver exogenous miR-145-5p to inhibit pancreatic ductal adenocarcinoma progression
Yixuan Ding, Feng Cao, Haichen Sun, Yecheng Wang, Shuang Liu, Yanchuan Wu. Qingye Cui, Wen Tong Mei, Fei Li
Cancer Letters
2019
ApplicationsCancerConcentration MeasurementExtracellular VesiclesPancreatic CancerSize MeasurementTechnical NanoparticlesVesicles
145-Exodeliverydrug deliveryexosomeshUCMSChuman umbilical cord mesenchymal stromal cellmicroRNAmiR-145miR-145-5pmiRNAmiRNA transferpancreatic ductal adenocarcinomaPDACtumor treatmentxenograft tumors
Full TextExosomes from endothelial progenitor cells improve outcomes of the lipopolysaccharide-induced acute lung injury
Yue Zhou, Pengfei Li, Andrew J. Goodwin, James A. Cook, Perry V. Halushka, Eugene Chang, Basilia Zingarelli and Hongkuan Fan
Critical Care
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
exosomal miRNAhsa-miR-206miR-206miRNA profilingneurotrophic factorschizophrenia biomarkersSCZSCZ biomarkersserum derived exosomes
Full TextExosomes from Human Gingiva-Derived Mesenchymal Stem Cells Combined with Biodegradable Chitin Conduits Promote Rat Sciatic Nerve Regeneration
Feng Rao, Dianying Zhang, Tengjiaozi Fang, Changfeng Lu, Bo Wang et al.
Stem Cells International
2019
ApplicationsExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
GMSC-derived exosomeshuman gingiva-derived mesenchymal stem cell exosomesnerve cell regenerationneurite outgrowthperipheral nerve repair
Full TextExosomes harbor B cell targets in pancreatic adenocarcinoma and exert decoy function against complement-mediated cytotoxicity
Michela Capello, Jody V. Vykoukal, Hiroyuki et al.
Nature Communications
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
B Cellexosome identificationexosome sortingexosomespancreatic ductal adenocarcinaomaPDACPDAC cell-derived exosomesPDAC-derived exosomestumor antigenstumor developmenttumor progression
Full TextExosomes Released from Rabies Virus-Infected Cells May be Involved in the Infection Process
Jingyu Wang, Fan Wu. Chuntian Liu, Wenwen Dai, Yawei Teng, Weiheng Su, Wei Kong, Feng Gao et al.
Virologica Sinica
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesViruses
exosome isolationexosomesrabiesrabies infectionrabies virus
Full TextExosomes secreted by mesenchymal stromal/stem cell-derived adipocytes promote breast cancer cell growth via activation of Hippo signaling pathway
Shihua Wang, Xiaodong Su, Meiqian Xu, Xian Xiao, Xiaoxia Li, Hongling Li, Armand Keating, and Robert Chunhua Zhao
Stem Cell Research & Therapy
2019
ApplicationsBreast CancerCancerConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
AdipocyteAdipocyte ExosomesBreast Cancercell-cell communicationexosomeshippo signaling pathwaymesenchymal stromal/stem celltumor development
Full TextExpression of Glypican 3 Is an Independent Prognostic Biomarker in Primary Gastro-Esophageal Adenocarcinoma and Corresponding Serum Exosomes
Mohammad Rahbari, Mathieu Pecqueux, Daniela Aust, Holger Stephan, Oliver Tiebel, Antonios Chatzigeorgiou, Torsten Tonn, Franziska Baenke et al.
Journal of Clinical Medicine
2019
BiomarkersCancerExosomesExtracellular VesiclesGastro-Esophageal AdenocarcinomaVesicles
diagnostic biomarkereGPC3Glypican 3GPC3
Full TextExpression profiles and potential functions of circular RNAs in extracellular vesicles isolated from radioresistant glioma cells
Mengjie Zhao, Jinjin Xu, Shanliang Zhong, Yuchi Liu, Hong Xiao, Liangyuan Geng, Hongyi Liu
Oncology Reports
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
circRNAcircular RNAEVsglioma radioresistanceNor-EVsradioresistanceU251 cells
Full TextExtracellular vesicle‑delivered miR‑505‑5p, as a diagnostic biomarker of early lung adenocarcinoma, inhibits cell apoptosis by targeting TP53AIP1
Hua Fang, Yutao Liu, Yaohong He, Yang Jiang, Yaping Wei, Han Liu, Yueqing Gong, Guangyu An
International Journal of Oncology
2019
ApplicationsBiomarkersCancerConcentration MeasurementExtracellular VesiclesLung AdenocarcinomaSize MeasurementTechnical NanoparticlesVesicles
EVsextracellular vesiclesLA biomarkerslung adenocarcinomalung adenocarcinoma biomarkerslung cancerlung cancer biomarkersmiR-382miR-486miR-505miRNAoncogenetargeted deliverytargeted EVs
Full TextExtracellular Vesicles and Ebola Virus: A New Mechanism of Immune Evasion
Michelle L. Pleet, Catherine DeMarino, Spencer W. Stonier, John M. Dye, Steven Jacobson, M. Javad Amanand Fatah Kashanchi
Viruses
2019
ApplicationsConcentration MeasurementExtracellular VesiclesReviewSize MeasurementTechnical Nanoparticles
cytokineEbola virusEBOV protein VP40exosomeextracellular vesiclesglycoproteinGPNPnucleoprotein
Full TextExtracellular vesicles derived from Echinococcus granulosus hydatid cyst fluid from patients: isolation, characterization and evaluation of immunomodulatory functions on T cells
Xiaojing Zhou, Wen Wang, Fang Cui, Chunli Shi, Yifei Ma, Yongzhuo Yu, Wei Zhao, Jiaqing Zhao
International Journal for Parasitology
2019
ApplicationsExtracellular VesiclesParasitesSize MeasurementVesicles
Echinococcus granulosusproteomicsT lymphocytes
Full TextExtracellular vesicles derived from Malassezia furfur stimulate IL-6 production in keratinocytes as demonstrated in in vitro and in vivo models
Yu-Jing Zhang, Yang Han, Yu-Zhe Sun, Hang-Hang Jiang, Min Liu, Rui-Qun Qi, Xing-Hua Gao
Journal of Dermatological Science
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
extracellular vesiclesIL-6inflammatory cutaneous disorderkeratinocytemalassezia furfurmalassezia pathogenesismalassezia therapynuclear factor-kappa Bskin inflammation
Full TextFli-1 transcription factor regulates the expression of caspase-1 in lung pericytes
Pengfei Li. Andrew J. Goodwin, James A. Cook. Perry V. Halushkad. Xian K. Zhang, Hongkuan Fan
Molecular Immunology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
caspase-1Fli-1friend leukemia viruslung pericytesOMVsouter membrane vesiclespericytes
Full TextGenome-Wide, Integrative Analysis Implicates Exosome-Derived MicroRNA Dysregulation in Schizophrenia
Yang Du Yun, Yu Yang Hu, Xiao-Wan Li, Ze-Xu Wei, Rui-Yuan Pan, Xue-Song Li, Guang-En Zheng, Xiao-Yan Qin, Qing-Shan Liu
Schizophrenia Bulletin
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesZeta Potential
Brain-derived Neurotrophic Factorexosomal miRNAexosomeshsa-miR-206microRNAmiR-206miRNAmiRNA profilingschizophreniaschizophrenia biomarkersSCZSCZ biomarkersserum derived exosomes
Full TextHuman Umbilical Cord Mesenchymal Stem Cell-Derived Extracellular Vesicles Inhibit Endometrial Cancer Cell Proliferation and Migration through Delivery of Exogenous miR-302a
Xin Li, Li li Liu, Ju lei Yao, Kai Wang and Hao Ai
Stem Cells International
2019
ApplicationsCancerConcentration MeasurementEndometrial CancerExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Anticancer Therapyendometrial cancerendometrial cancer therapyengineered extracellular vesiclesEV engineeringhUCMSChuman umbilical cord mesenchymalstem cellsmicroRNAmiR-302amiRNAstem cellstumor suppressor miRNAtumor-targeting drug delivery
Full TextHuman umbilical cord mesenchymal stem cells derived exosomes exert antiapoptosis effect via activating PI3K/Akt/mTOR pathway on H9C2 cells
Hui Liu, Xiaolu Sun, Xuhe Gong, Guogan Wang
Journal of Cellular Biochemistry
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
ApoptosisAutophagycardiovascular diseasesCVDexosomesnull-nullPI3K/Akt/mTOR pathwayumbilical cord mesenchymal stem cells
Full TextHypoxia preconditioned renal tubular epithelial cell-derived extracellular vesicles alleviate renal ischaemia-reperfusion injury mediated by the HIF-1α/Rab22 pathway and potentially affected by microRNAs
Lei Zhang, Han Liu, Kai Xu, Zhixin Ling, Yeqing Huang, Qiang Hu, Kai Lu, Chunhui Liu et al.
International Journal of Biological Sciences
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
extracellular vesiclesHK2 cellsHPCHPC EVshypoxiaischaemiaischaemia-reperfusion injuryischaemic preconditioningkidney diseasemicroRNArenal injury
Full TextIdentification of a potential exosomal biomarker in spinocerebellar ataxia Type 3/Machado–Joseph disease
Xiaocan Hou, Xuan Gong, Longbo Zhang, Tianjiao Li, Hongyu Yuan,YueXie,YunPeng, Rong Qiu,KunXia, Beisha Tang & Hong Jian
Epigenomics
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Age at OnsetAxon Guidanceclinical classificationendoplasmic reticulumexosomal biomarkersexosomesmiRNAsSCA3/MJspinocerebellar ataxia type 3 (SAC3)
Full TextIncreased urinary exosomal microRNAs in children with idiopathic nephrotic syndrome
Tingting Chen, Cheng Wang, Hanqing Yu, Meng Ding, Cuiping Zhang, Xiaolan Lu, Chen-Yu Zhang, Chunni Zhang
eBioMedicine
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
exosomesillumina sequencing via synthesiskidney diseasekidney disease diagnosismicroRNAmiRNAnon-invasive biomarkerspaediatric idiopathic nephrotic syndromeRT-qPCRurinary exosomes
Full TextIndividual Immune-Modulatory Capabilities of MSC-Derived Extracellular Vesicle (EV) Preparations and Recipient-Dependent Responsiveness
Lambros Kordelas, Esther Schwich, Robin Dittrich, Peter A. Horn, Dietrich W. Beelen, Verena Börger, Bernd Giebel and Vera Rebmann
International Journal of Molecular Sciences
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Acute InflammationCD4CD45RACD8EVsextracellular vesiclesgraft-versus-host-diseaseGvHDinflammation treatmentinflammation-associated disordersmesenchymal stem cellsmesenchymal stromal cellsMSCMSC-derived EVsMSC-EVssurface markers
Full TextInhibition of multiple myeloma‑derived exosomes uptake suppresses the functional response in bone marrow stromal cell
Yongjiang Zheng, Chenggong Tu, Jingwen Zhang, Jinheng Wang
International Journal of Oncology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
BMSCBone Marrow Stromal CellsexosomesMM cell-derived exosomesMM cellsMM treatmentmultiple myeloma cellsmultiple myeloma treatment
Full TextInnate extracellular vesicles from melanoma patients suppress β-catenin in tumor cells by miRNA-34a
Jung-Hyun Lee, Jochen Dindorf, Martin Eberhardt, Xin Lai, Christian Ostalecki, Nina Koliha, Stefani Gross,Katja Blume, Heiko Bruns, Stefan Wild, Gerold Schuler, Julio Vera, Andreas S Baur
Life Science Alliance
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
extracellular vesiclesmelanomamelanoma treatmentpEVsplasma-derived EVstumor development
Full TextMacrophage-Derived miRNA-Containing Exosomes Induce Peritendinous Fibrosis after Tendon Injury through the miR-21-5p/Smad7 Pathway
Haomin Cui, Yu He, Shuai Chen Deming Zhang, Yaling Yu, Cunyi Fan
Molecular Therapy - Nucleic Acids
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Bone Marrow Macrophageexosomeexosomeshand surgerymacrophage-derived exosomesmicroRNAmiR-21miR-21-5pmiRNAperitendinous adhesionperitendinous fibrosisperitendinous tissue fibrosisSmad7tendon adhesion preventiontendon adhesion treatmenttendon injurytendon-healing
Full TextMatrix stiffness regulates microvesicle-induced fibroblast activation
Schwager SC, Bordeleau F, Zhang J, Antonyak MA, Cerione RA, Reinhart-King C.
American Journal of Physiology. Cell Physiology
2019
ApplicationsCancerConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesZeta Potential
cancer therapycancer treatmentcell contractilityextracellular matrixextracellular secreted vesicleextracellular vesiclesmechanotransductiontraction force
Full TextMiRNA profiling of plasma-derived exosomes from dairy cows during gestation
Gan Zhao, Shuai Guo, Kangfeng Jiang, Tao Zhang, Haichong Wu, Changwei Qiu, Ganzhen Deng
Theriogenology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
exosomesmaternal-fetal communicationmaternal-fetal immune responsemiRNAsplasma derived exosomesplasma-derived EVsplasma-derived exosomal miRNApregnancyRNA-seq
Full TextOptimisation of imaging flow cytometry for the analysis of single extracellular vesicles by using fluorescence-tagged vesicles as biological reference material
André Görgens, Michel Bremer, Rita Ferrer-Tur, Florian Murke, Tobias Tertel, Peter A. Horn, Sebastian Thalmann, Joshua A. Welsh, Christine Probst, Coralié Guerin, Chantal M. Boulanger, Jennifer C. Jones, Helmut Hanenberg, Uta Erdbrügger, Joanne Lannigan, Franz L. Ricklefs, Samir El-Andaloussi and Bernd Giebel
Journal of Extracellular Vesicles
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementSmall Extracellular VesiclesTechnical NanoparticlesVesicles
Antibody LabellingCD63ev characterizationflow cytometrysEVssingle EV detectionsmall EV characterizationsmall EV identificationsmall EVssubpopulations
Full TextProteomic Analysis on Exosomes Derived from Patients’ Sera Infected with Echinococcus granulosus
Wen Wang, Xiaojing Zhou, Fang Cui, Chunli Shi, Yulan Wang, Yanfei Men, Wei Zhao and Jiaqing Zhao
The Korean Journal of Parasitology
2019
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesParasitesSize MeasurementVesicles
Echinococcus granulosusproteomics
Full TextRat sinus mucosa‐ and periosteum‐derived exosomes accelerate osteogenesis
Ruinan Sun, Shuyu Xu, Zuolin Wang
Journal of Cellular Physiology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Alveolar Bone AugmentationBone RegenerationBone Tissue EngineeringexosomesGBRguided bone regenerationosteogenesisperiosteumsinus mucosa
Full TextRegulation of exosomes secretion by low-intensity pulsed ultrasound in lung cancer cells
Qingyu Zeng, Shibin Hong, Xue Wang, Yirui Cheng, Junfeng Sun, Weiliang Xia
Experimental Cell Research
2019
ApplicationsCancerConcentration MeasurementExtracellular VesiclesLung CancerSize MeasurementTechnical NanoparticlesVesicles
exosome-related diseasesexosomesLIPUSLIPUS/exosome interactionLow-intensity pulsed ultrasoundmTOR
Full TextSodium Taurocholate Stimulates Campylobacter jejuni Outer Membrane Vesicle Production via Down-Regulation of the Maintenance of Lipid Asymmetry Pathway
Hong Cheng, Jing-Hao Fan, Lin-Ping Zhao, Gui-Ling Fan, Rong-Rong Zheng, Xiao-Zhong Qiu, Xi-Yong Yu, Shi-Ying Li, Xian-Zheng Zhang
Biomaterials
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Bile SaltsCampylobactermaintenance of lipid asymmetry pathwayMlaAouter membrane vesiclesvirulence
Full TextSubmicron Particle Focusing and Exosome Sorting by Wavy Microchannel Structures within Viscoelastic Fluids
Yinning Zhou, Zhichao Ma, Mahnoush Tayebi, Ye Ai
Analytical Chemistry
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
EV purificationexosome purificationexosome sortingexosomeshigh-throughput sortingparticle sortingreverse wavy channel
Full TextUpregulation of miR-489-3p and miR-630 inhibits oxaliplatin uptake in renal cell carcinoma by targeting OCT2.
Lu Chen, Le Chen, Zhiyuan Qin, Jinxiu Lei, Sheng Ye, Kui Zeng Hua Wang, Meidan Ying, Jianqing Gao, Su Zeng, Lushan Yu
Acta Pharmaceutica Sinica B
2019
ApplicationsCancerConcentration MeasurementExtracellular VesiclesRenal Cell CarcinomaSize MeasurementTechnical NanoparticlesVesicles
epigenetic regulationmicroRANmiR-489miR-630miRNAOCT2OCT2 expressiononcotherapyoxaliplatinoxaliplatin resistanceRCCRCC treatmentrenal cancer treatmentrenal cell carcinoma
Full TextVery Long-Chain C24:1 Ceramide Is Increased in Serum Extracellular Vesicles with Aging and Can Induce Senescence in Bone-Derived Mesenchymal Stem Cells
Andrew Khayrullin, Priyanka Krishnan, Luis Martinez-Nater, Bharati Mendhe, Sadanand Fulzele, Yutao Liu, Julie A. Mattison and Mark W. Hamrick
Cells
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
AgingApoptosiscardiovascular healthcell senescencememory impairmentserum derived exosomesvery long-chain ceramides
Full TextAnchor peptide captures, targets, and loads exosomes of diverse origins for diagnostics and therapy
X Gao, N Ran, X Dong, B Zuo, R Yang, Q Zhou et al.
Science Translational Medicine
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
cargoCD63CP05dystrophin expressionexosomal anchor peptideexosome captureexosome engineeringexosome modificationmuscular dystrophypeptidesplicing
Full TextAntiretroviral Drugs Alter the Content of Extracellular Vesicles from HIV-1-Infected Cells
C DeMarino, ML Pleet, M Cowen, RA Barclay et al.
Scientific Reports
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesViruses
cART-altered EV contentCD63CD81CD9DemeclocyclineDoxycyclineextracellular vesiclesFDA-approved drugsHIV-1MethacyclineMinocyclinenon-coding viral RNAOxycyclineOxytetracyclineSCRT pathwayTAR RNtetracyclineVPS4
Full TextBeta cell extracellular vesicle miR-21-5p cargo is increased in response to inflammatory cytokines and serves as a biomarker of type 1 diabetes
Lakhter A, Pratt RE, Moore RE, Doucette KK1, Maier BF, DiMeglio LA, Sims EK
Diabetologia
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Beta Cell Signal TransductioncalreticulincaspaseCD63CD9cell linescytokinediabetes biomarkerdiabetes predictiondiabetes preventionEVsexomiRexomiR-21extracellular vesicleshumanmicroRNAmiR-21miRNAtype 1 diabetes
Full TextCircular RNA expression in extracellular vesicles isolated from serum of patients with endometrial cancer
Hanzi Xu, Zhen Gong, Yang Shen, Yichen Fang& Shanliang Zhong .
Epigenomics
2018
ApplicationsCancerConcentration MeasurementEndometrial CancerExosomesExtracellular VesiclesSize MeasurementVesicles
Circular RNAsdifferential circRNA expressionendometrial cancerexosomesextracellular vesiclesmiRNAstage III adenocarcinoma
Full TextCommunication of prostate cancer cells with bone cells via extracellular vesicle RNA; a potential mechanism of metastasis.
C. Probert, T. Dottorini, A. Speakman, S. Hunt, T. Nafee, A. Fazeli, S. Wood, J. E. Brown & V. James
Oncogene
2018
ApplicationsCancerConcentration MeasurementExtracellular VesiclesProstate CancerSize MeasurementTechnical NanoparticlesVesicles
cancer preventioncancer treatmentcell-cell communicationEV cargoEVsextracellular vesiclesPCaprostate cancerprostate cancer treatmenttargeted EVstumor derived exosomes
Full TextDetailed analysis of the plasma extracellular vesicle proteome after separation from lipoproteins
Nasibeh Karimi, Aleksander Cvjetkovic, Su Chul Jang, Rossella Crescitelli, Mohammad Ali Hosseinpour Feizi, Rienk Nieuwland, Jan Lötvall, Cecilia Lässer.
Cellular and Molecular Life Sciences
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Apo-A1CD63CD81CD9Density cushionexosomesextracellular vesiclesFlotillin-1LipoproteinsMass spectrometryPlasmaproteomicsSerumSize-exclusion chromatography
Full TextDevelopment of MSC-NTF cell exosomes for the treatment of neurodegenerative diseases
Kaspi, Haggai; Semo, Jonathan; Abramov, Natalie; Aricha, Revital.
Journal of Extracellular Vesicles
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Anti-inflammatoryBBBBlood–Brain BarrierBone Marrow Derived Mesenchymal Stem Cellsdrug deliveryEV cargomicroRNAmiRNAMSCMSC-derived EVsMSC-EVsMSC-NTF cellsneurodegenerative diseasesneuronal growthnon-invasive therapytargeted deliverytargeted EVstargeted exosomes
Full TextDiet-resistant obesity is characterized by a distinct plasma proteomic signature and impaired muscle fiber metabolism
A B Thrush, G Antoun, M Nikpay, D A Patten, C DeVlugt, J-F Mauger, B L Beauchamp, P Lau, R Reshke, É Doucet, P Imbeault, R Boushel, D Gibbings, J Hager, A Valsesia, R S Slack, O Y Al-Dirbashi, R Dent, R McPherson & M-E Harper.
International Journal of Obesity
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Diet resistanceexosomesfasting conditionshigh fat mealHSP72muscle metabolismobese diet-resistanceobesityoxidative stressplasma proteomicsROS
Full TextDifferential fluorescence nanoparticle tracking analysis for enumeration of the extracellular vesicle content in mixed particulate solutions
Pachler, Karin; Desgeorges, Alexandre; Folie, Christina; Mayr, Magdalena; Binder, Heide-Marie; et al.
Journal of Extracellular Vesicles
2018
ApplicationsConcentration MeasurementExtracellular VesiclesF-NTAFluorescenceSize MeasurementTechnical NanoparticlesVesicles
AlexaFluor 488Antibody LabellingAntibody Stainingev characterizationEV quantificationEVsextracellular vesiclesf-NTAfluorescence NTAfluorescent labellingfNTAUCMSC-derived EVsumbilical cord mesenchymal stromal cells-derived EVs
Full TextDPTIP, a newly identified potent brain penetrant neutral sphingomyelinase 2 inhibitor, regulates astrocyte-peripheral immune communication following brain inflammation
Camilo Rojas , Elena Barnaeva, Ajit G. Thomas, Xin Hu et al.
Scientific Reports
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Astrocyte-derived EVsBrain InflammationBrain InjurycytokineDPTIPEVsmicroRNAmiRNAneutral sphingomyeliinase 2nSMase2nSMase2 inhibition
Full TextEbola Virus VP40 Modulates Cell Cycle and Biogenesis of Extracellular Vesicles
Michelle L Pleet, James Erickson, Catherine DeMarino, Robert A Barclay, Maria Cowen, Benjamin Lepene, Janie Liang, Jens H Kuhn, Laura Prugar, Spencer W Stonier, John M Dye, Weidong Zhou, Lance A Liotta, M Javad Aman, Fatah Kashanchi
The Journal of Infectuos Diseases
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesViruses
Apoptosiscell cycleebola treatmentEbola virusEBOVEBOV pathogenesiEBOV treatmentEV biogenesisEVsextracellular vesiclesmonocyte apoptosispoptosis; cell cycleT cell apoptosisVP40
Full TextEffect of plasma-derived extracellular vesicles on erythrocyte deformability in polymicrobial sepsis
Kumar Subramani, Srikrishnan P. Raju, Xiaogang Chua, Marie Warren, Chirayu D. Pandyab, Nasrul Hoda, Sadanand Fulzele, Raghavan Raju
International Immunopharmacology
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
erythrocytesEV cargoexosomesextracellular vesiclesmicroRNAmiRNAmiRNA cargomultiple organ dysfunctionRBCsred blood cellsrheologysepsistissue hypoxiatissue perfusion
Full TextEmerging role of extracellular vesicles in musculoskeletal diseases
Murphy C, Withrow J, Hunter M, Liu Y, Tang YL, Fulzele S, Hamrick MW.
Molecular Aspects of Medicine
2018
Extracellular VesiclesVesicles
Bone MetabolismEV-derived miRNAexosomesfibroblast growth factorinsulin-like growth factor-1microRNAmicrovesiclesmuscular dystrophymyoblastmyotubeosteoarthritisosteoporosisrheumatoid arthritistransforming growth factor-beta1vascular endothelial growth factor
Full TextEpigenetic modifications in hyperhomocysteinemia: potential role in diabetic retinopathy and age-related macular degeneration
Elmasry K, Mohamed R, Sharma I, Elsherbiny NM, Liu Y, Al-Shabrawey M, Tawfik A.
Oncotarget
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Age-related Macular DegenerationAngiogenesisBlood Retinal Barrierdiabetic retinopathyepigenetic modificationsGerotargethyperhomocysteinemiahypoxiainflammationmiRNAoxidative stress
Full TextEutopic stromal cells of endometriosis promote neuroangiogenesis via exosome pathway
Huihui Sun, Dong Li, Ming Yuan ,Qiuju Li, Ni li, Guoyun Wang
Biology of Reproduction
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
endometrial stromal cells-derived exosomesendometriosisendometrium-derived exosomesESC-derived exosomesexosomesGW4869neuroangiogenesisneuroangiongenesis
Full TextExoAPP: Exosome-Oriented, Aptamer Nanoprobe-Enabled Surface Proteins Profiling and Detection
Dan Jin, Fan Yang, Yulin Zhang, Li Liu, Yujuan Zhou, Fubing Wang, and Guo-Jun Zhang
Analytical Chemistry
2018
ApplicationsCancerConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
cancer diagnosisExoAppexosome-oriented aptamer nanoprobeexosomestargeted EVstumor derived exosomes
Full TextExosomal circRNA_100284 from arsenite-transformed cells, via microRNA-217 regulation of EZH2, is involved in the malignant transformation of human hepatic cells by accelerating the cell cycle and promoting cell prolife
X Dai, C Chen, Q Yang, J Xue, X Chen, B Sun, F Luo et al.
Cell Death & Disease
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
ArseniteArsenite-induced Carcinogenesiscell cyclecirculting RNAExosomal circRNA_100284exosomeslivermalignant transformationmiRNAproliferation
Full TextExosomal microRNA-21 derived from bronchial epithelial cells is involved in aberrant epithelium-fibroblast cross-talk in COPD induced by cigarette smoking
Xu H, Ling M, Xue J, Dai X, Sun Q, Chen C, Liu Y, Zhou L, Liu J, Luo F, Bian Q, Liu Q.
Theranostics
2018
ApplicationsConcentration MeasurementDiagnosticsExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Bronchial Epithelium-fibroblast Communicationcell-cell communicationchronic obstructive pulmonary diseasecigarette smoke induced COPDCOPDCOPD diagnosisCOPD treatmentCS-induced COPDEVs intercelllar communicationexosomesmicroRNAmiR-21miRNA
Full TextExosome markers associated with immune activation and oxidative stress in HIV patients on antiretroviral therapy
S Chettimada, DR Lorenz, V Misra, ST Dillon et al.
Scientific Reports
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesViruses
CD63CD81CD9CRPD14exosome cargoHIVHLA-AHLA-Bimmunological markersimmunomodulationoxidative stressplasma exosome
Full TextExosome-Derived Dystrophin from Allograft Myogenic Progenitors Improves Cardiac Function in Duchenne Muscular Dystrophic Mice
Su X, Jin Y, Shen Y, Ju C, Cai J, Liu Y, Kim IM, Wang Y, Yu H, Weintraub NL, Jiang M, Tang Y.
Journal of Cardiovascular Translational Research
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Allogenic Exosomal Deliverycardiomyopathycardiomyopathy treatmentDMDDMD treatmentdrug deliveryduchenne muscular dystrophydystrophinexosomesMPC derived exosomesMPC-exomyogenic progenitor cellstargeted delivery
Full TextExosomes from asbestos-exposed cells modulate geneexpression in mesothelial cells
Phillip Munson, Ying-Wai Lam, Julie Dragon, Maximilian MacPherson and Arti Shukla.
The FASEB Journal
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Asbestos Exposure MesotheliomaCD81cell cyclechromosomal stabilityextracellular vesiclesgene expressionhuman mesothelia cellsproteomic cargoproteomicsTGFbtumorigenesis
Full TextExosomes from Suxiao Jiuxin Pill-treated cardiac mesenchymal stem cells decrease H3K27 demethylase UTX expression in mouse cardiomyocytes in vitro
Xiao-fen Ruan, Yong-jun Li, Cheng-wei Ju, Yan Shen, Wei Lei, Can Chen, Yang Li, Hong Yu, Yu-tao Liu, Il-man Kim, Xiao-long Wang, Neal L Weintraub & Yao-liang Tang.
Acta Pharmacologica Sinica
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Borneolcardiac mesenchymal stem cellsCD63CD81epigenetic regulationexosomesH3K27 demethylasesH3K27 methylasesPCNASuxiao Jiuxin pilltetramethylpyrazinetraditional chinese medicineUTX
Full TextExosomes originating from MSCs stimulated with TGF-β and IFN-γ promote Treg differentiation
Zhang, Q., Fu, L., Liang, Y., Guo, Z., Wang, L., Ma, C. and Wang, H.
Journal of Cellular Physiology
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
CD63CD81CD9cytokinesexosomesIDOIFN-γIL-10immune modulationimmunosuppressive activitymesenchymal stem cellsMSC-exTGF-βtherapeutic effectumbilical cord-derived MSCs
Full TextExtracellular vesicle-carried Jagged-1 inhibits HUVEC sprouting in a 3D microenvironment
Tan, E., Asada, H.H. & Ge, R.
Angiogenesis
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
AngiogenesisDLL4endothelial cellsextracellular vesiclesHUVECJA1-ICDJAG1NOTCH signallingproliferation
Full TextExtracellular vesicle-mediated long-range communication in stressed retinal pigment epithelial cell monolayers
Shah N, Ishii M, Brandon C, Ablonczy Z, Cai J, Liu Y, Chou CJ, Rohrer B
Biochimica et biophysica acta. Molecular basis of disease
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Age-related Macular DegenerationEVsexosomesextracellular vesiclesHDAC6retinal pigment epitheliumRPERPE dysfunctiontight junctions
Full TextExtracellular vesicles or free circulating DNA: where to search for BRAF and cKIT mutations?
Jennifer Klump MD, Ulrike Phillipp, Marie Follo PhD, Anna Eremin MSc, Hannes Lehmann MD, Sigrun Nestel, Nikolas von Bubnoff Prof. Dr., Irina Nazarenko PD
Nanomedicine: Nanotechnology, Biology and Medicine
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
BRAFBRAF600Ecell-free circulating DNAcKITcKITD816VEV DNA contentextracellular vesiclesImatinibLiquid biopsymelanomapredictive biomarkersTherapy monitoringVemurafenib
Full TextFetal Bovine Serum-Derived Extracellular Vesicles Persist within Vesicle-Depleted Culture Media
Brandon M. Lehrich , Yaxuan Liang, Pooya Khosravi , Howard J. Federoff and Massimo S. Fiandaca
International Journal of Molecular Sciences
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Astrocyte Cell GrowthCMculture mediaEV isolationEV purificationEV-depletion methodsEVsexosomesextracellular vesiclesfetal bovine serumnanoparticle tracking analysis
Full TextGlucocerebrosidase deficiency promotes protein aggregation through dysregulation of extracellular vesicles
Ruth E. Thomas , Evelyn S. Vincow, Gennifer E. Merrihew, Michael J. MacCoss, Marie Y. Davis, Leo J. Pallanck
PLOS GENETICS
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
EVsextracellular vesiclesGBAglucocerebrosidase deficiencyglucosylceramidase betaneurodegenerative diseases
Full TextHuman Mesenchymal Stromal Cell-Derived Extracellular Vesicles Modify Microglial Response and Improve Clinical Outcomes in Experimental Spinal Cord Injury
Katherine A. Ruppert, Tin T. Nguyen, Karthik S. Prabhakara, Naama E. Toledano Furman, Amit K. Srivastava, Matthew T. Harting, Charles S. Cox Jr. & Scott D. Olson.
Scientific Reports - Nature
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
CD107CD63CD81CD9COX2/PGE2 pathwaycytokinesextracellular vesiclesinflammation attenuationinflammatory effectslocomotor recoverymesenchymal stromal cellsMicroglial ResponseSpinal Cord Injury
Full TextIntegrated Nanoscale Deterministic Lateral Displacement Arrays for Separation of Extracellular Vesicles from Clinically-Relevant Volumes of Biological Samples
Joshua T. Smith, Benjamin H. Wunsch, Navneet Dogra et al.
Lab on a Chip
2018
ApplicationsBiomarkersConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
diagnosisdiagnostic biomarkerearly-stage disease diagnosisEV isolationEV purificationEV separationEVsextracellular vesiclesnanoDLDnanoscale deterministic lateral displacementtargeted deliverytargeted EVstreatment monitoring
Full TextLack of chitin synthase genes impacts capsular architecture and cellular physiology in Cryptococcus neoformans
J Rodrigues, CL Ramos, S Frases, RMC Godinho et al.
The Cell Surface
2018
ApplicationsConcentration MeasurementExtracellular VesiclesFungiSize MeasurementVesicles
Antifungal Developmentchitin metabolismchitin synthase geneschitinaseCryptococcus neoformansdeletionextracellular vesiclesglucuronoxylomannantargetvirulence
Full TextLarge-scale, cross-flow based isolation of highly pure and endocytosis-competent extracellular vesicles
Ryan P. McNamara, Carolina P. Caro-Vegas, Lindsey M. Costantini
Journal of Extracellular Vesicles
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
capto corecross-flowEV isolationEV purificationEVsextracellular vesiclesindustrial scale EV purificationindustrial scale EV-isolationtangential flow
Full TextLipopolysaccharide-Preconditioned Periodontal Ligament Stem Cells Induce M1 Polarization of Macrophages through Extracellular Vesicles
Hyejong Kang, Myung-Ju Lee, Sang June Park and Myung-Shin Lee
International Journal of Molecular Sciences
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
EVsextracellular vesiclesgum inflammationgum inflammation therapyM1 polarization of macrophagesmacrophagePDLSCperiodonitis therapyperiodontal diseaseperiodontal ligament stem cellperiodontitspolarization
Full TextMesenchymal stromal cell exosome–enhanced regulatory T-cell production through an antigen-presenting cell–mediated pathway
Bin Zhang Ronne Wee Yeh Yeo, Ruenn Cha iLai, Eugene Wei Kian Sim, Keh Chuang Chin, Sai Kiang Lim.
Cytotherapy
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Antigen-presenting CellAPC PathwaycargoCD81CD9exosomeexosome-mediated Treg productiongraft-versus-host-diseasemesenchymal stromal cellregulatory T cells
Full TextMetabolic reprogramming of stromal fibroblasts by melanoma exosome microRNA favours a pre-metastatic microenvironment
Shin La Shu, Yunchen Yang, Cheryl L. Allen, Orla Maguire, Hans Minderman et al.
Scientific Reports
2018
ApplicationsCancerConcentration MeasurementExtracellular VesiclesMelanomaSize MeasurementTechnical NanoparticlesVesicles
cancer diagnosiscancer metastasiscancer therapyexosomesextracellular acidificationHADFHMEXhuman adult dermal fibroblastshuman melanoma-derived exosomesmetastasis developmentmetastasis promotionmicroRNAmiR-155miR-210miRNAmiRNA cargo
Full TextNef Secretion into Extracellular Vesicles or Exosomes Is Conserved across Human and Simian Immunodeficiency Viruses
Ryan P. McNamara, Lindsey M. Costantini, T. Alix Myers, Blake Schouest, Nicholas J. Maness, Jack D. Griffith, Blossom A. Damania, Andrew G. MacLean, Dirk P. Dittmer.
mBio
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesiclesViruses
AIDSCD4CD63CD81exosomesextracellular vesiclesFlotillinHIVlentivirusesmicrovesiclesNefSIV
Full TextPhenotyping of EVs by multiwavelength fluorescence nanoparticle tracking Analysis
Helmbrecht, Clemens
Journal of Extracellular Vesicles
2018
ApplicationsConcentration MeasurementExtracellular VesiclesF-NTAFluorescenceSize MeasurementTechnical NanoparticlesVesicles
EV phenotypingf-NTAfNTAmultiwavelength NTAnanoparticle tracking analysisNTAsingle EV detectionsingle particle analysissingle particle detectionsubpopulations
Full TextPhenotyping of EVs by multiwavelength fluorescence nanoparticle tracking Analysis
Bremer, Michel; Ferrer-Tur, Rita; Görgens, André; Börger, Verena; Horn, Peter A; et al.
Journal of Extracellular Vesicles
2018
ApplicationsConcentration MeasurementExtracellular VesiclesF-NTAFluorescenceSize MeasurementTechnical NanoparticlesVesicles
EVsextracellular vesiclesf-NTAflow cytometryfNTAnanoparticle characterizationnanoparticle measurementsingle EV detectionsingle particle analysissingle particle detection
Full TextPlacental exosome-mediated Bta-miR-499-Lin28B/let-7 axis regulates inflammatory bias during early pregnancy
G Zhao, C Yang, J Yang, P Liu, K Jiang, A Shaukat et al.
Cell Death & Disease
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Abnormal Inflammatory BiasBta-miR-499CD63CD9exosomesLin28B/let-7 axismiRNANF-κBplacentapregnancy
Full TextProbing the role of myofibroblast-derived extracellular vesicles in cancer
Higginbotham, Samuel J; Hunt, Stuart; Lambert, Daniel W
Journal of Extracellular Vesicles
2018
ApplicationsCancerConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
CAFcancer associated fibroblastscancer cell-derived EVscancer progressionEV cargoEVsexosome cargoextracellular vesiclesfibroblast derived exosomesmicroRNAmiRNAmyofibroblast-derived exosomesmyofibroblastsNOFnormal oral fibroblastssolid tumortumor biomarkerstumor diagnosistumor prognosis
Full TextProgress in separation of extracellular vesicles from brain tissue ofhuman, macaque and mouse
Yiyao Huang; Laura J. Vella; Vasiliki Machairaki; Linzhao Cheng; Andrew F. Hill; Lei Zheng; Kenneth Witwer
Journal of Extracellular Vesicles
2018
ApplicationsConcentration MeasurementExtracellular VesiclesTechnical NanoparticlesVesicles
Brain-derived EVscentral nervous system diseasesCNS diseasesEV isolationEV purificationEV separationexosomal biomarkershuman brain tissue-derived EVs
Full TextPurification of extracellular vesicles from plasma by heparin-coated magnetic beads
Yiyao Huang; Dillon C. Muth; Lei Zheng; Kenneth W. Witwer
Journal of Extracellular Vesicles
2018
ApplicationsConcentration MeasurementExtracellular VesiclesTechnical NanoparticlesVesicles
EV isolationEV purificationEVsexosomesheparin-coated magnetic beadsosomesplasma derived exosomesplasma-derived EVs
Full TextReceptor-Ligand Interaction Mediates Targeting of Endothelial Colony Forming Cell-derived Exosomes to the Kidney after Ischemic Injury
Jose L. Viñas, Matthew Spence, Alex Gutsol, William Knoll, Dylan Burger, Joseph Zimpelmann, David S. Allan2 & Kevin D. Burns
Scientific Reports
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
ECFC-derived exosomesendothelial colony forming cell-derived exosomesischaemia-reperfusion injuryischemic kidneyischemic kidney injurymicroRNAmiR-486miRNAtargeted EVs
Full TextRegulation of Macrophage Activation and Polarization by HCC-Derived Exosomal lncRNA TUC339
Xue Li, Yi Lei, Miao Wu and Nan Li
International Journal of Molecular Sciences
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
exosomes; macrophage; lncRNA; inflammation; polarizationHCCHCC-derived exosomeshepatocellular carcinomahepatocellular carcionma cell-derived exosomeslong non-coding RNAtargeted deliverytargeted EVstumor derived exosomestumor-immune cell interaction
Full TextRemote ischaemic preconditioning increases serum extracellular vesicle concentrations with altered micro‐RNA signature in CABG patients
Ulrich H. Frey, Marina Klaassen, Crista Ochsenfarth, Florian Murke, Matthias Thielmann, Eva Kottenberg, Petra Kleinbongard, Stefanie Klenke, Andrea Engler, Gerd Heusch, Bernd Giebel. Jürgen Peters
Anaesthesiologica Scandinavica
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
CABGcardioprotectioncardiovascular diseasescoronary aretery bypass surgeryEVsextracellular vesiclesmicroRNAmiRNAmyocardial ischaemia injurymyocardial reperfusion injuryremote ischaemic preconditioningRIPC
Full TextSalvianolic acid A inhibits tumor-associated angiogenesis by blocking GRP78 secretion
Yang, Y., Zhang, L., La, X. et al. Jürgen Peters
Naunyn-Schmiedeberg's Archives of Pharmacology
2018
ApplicationsCancerConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
AngiogenesisAntiangiogenesisdrug resistanct cancer cellsexosomesglucose-regulated protein 78GRP78interactionSAAsalvianolic acid Asalvianolic acidATCMtraditional chinese medicinetumor developmenttumor-secreted GRP78
Full TextSoluble E-cadherin promotes tumor angiogenesis and localizes to exosome surface
MKS Tang, PYK Yue, PP Ip, RL Huang, HC Lai et al.
Nature Communications
2018
ApplicationsCancerConcentration MeasurementExosomesExtracellular VesiclesOvarian CancerSize MeasurementVesicles
AsciteBiomarkerE-cadherinexosomesHUVECNFκB signalingovarian cancersE-cadtumor angiogenesisβ-catenin
Full TextSuxiao Jiuxin pill promotes exosome secretion from mouse cardiac mesenchymal stem cells in vitro
Xiao-fen Ruan, Cheng-wei Ju, Yan Shen, Yu-tao Liu, Il-man Kim, Hong Yu, Neal Weintraub, Xiao-long Wang & Yao-liang Tang.
Acta Pharmacologica Sinica
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Acute Myocardial IschemiaBorneolcardiac mesenchymal stem cellsCD63exosomesGTPasenSMase2Rab27Suxiao Jiuxin pilltetramethylpyrazinetraditional chinese medicineTsg101
Full TextSynovial fluid-derived exosomal lncRNA PCGEM1 as biomarker for the different stages of osteoarthritis
Ye Zhao, Juan Xu
International Orthopaedics
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
exosomal biomarkersexosomal lncRNAexosomeslate-stage OAlncRNAlong non-coding RNAOAosteoarthritisosteoarthritis diagnosism early OA
Full TextThe chemokine receptor CCR1 is identified in mast cell-derived exosomes
Yuting Liang, Longwei Qiao, Xia Peng, Zelin Cui, Yue Yin, Huanjin Liao, Min Jiang, Li Li
American Journal of Translational Research
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Bioinformatical AnalysisCCR1CD9differentially expressed genesexosomesimmune regulationKITmast cell-derived exosomesMast cellsTGFBR1TLR9TPSAB1TPSB2
Full TextThe effect of storage temperature on the biological activity of extracellular vesicles for the complement system
Sang June Park, Hyungtaek Jeon. Seung-Min Yoo, Myung-Shin Lee.
In Vitro Cellular & Developmental Biology - Animal
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
Biological Activityclinical applicationcomplement systemEV markersexpressionextracellular vesiclesstorage condition
Full TextThe Role of Arrestin Domain-Containing 3 in Regulating Endocytic Recycling and Extracellular Vesicle Sorting of Integrin β4 in Breast Cancer
Young Hwa Soung, Shane Ford, Cecilia Yan and Jun Chung
Cancers
2018
ApplicationsBreast CancerCancerConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
ARRDC3Arrestin Domain-containing 3Breast CancerBreast Cancer Therapycancer therapyendocytic recyclingEVsextracellular vesiclesintegrin β4ITG 4TNBCtriple-negative breast cancerubiquitination
Full TextThe Serum Exosome Derived MicroRNA-135a, -193b, and-384 Were Potential Alzheimer’s Disease Biomarkers
YANG Ting Ting, LIU Chen Geng, GAO Shi Chao, ZHANG Yi, WANG Pei Chang.
Biomedical and Environmental Sciences
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Alzheimer’s DiseaseAPP/PS1 Transgenic MiceBACE-1BiomarkerCD63CD9differential expressionexosomal miRNAexosomehippocampusmicroRNAmiR-135amiR-193bmiR-384
Full TextTissue factor mediates microvesicles shedding from MDA-MB-231 breast cancer cells
AM da R Rondon, VH de Almeida, T Gomes et al.
Biochemical and Biophysical Research Communications
2018
ApplicationsBreast CancerCancerConcentration MeasurementExtracellular VesiclesMicrovesiclesSize MeasurementVesicles
Breast Cancercofilincytoskeletonextracellular vesiclesfasudilLIMKMDA-MB-231microvesiclesRhoA expressionROCKsilencingTissue factor
Full TextTNFα and IL-1β modify the miRNA cargo of astrocyte shed extracellular vesicles to regulate neurotrophic signaling in neurons
Amrita Datta Chaudhuri, Raha M. Dastgheyb, Seung-Wan Yoo, Amanda Trout, C. Conover Talbot Jr, Haiping Hao, Kenneth W. Witwer & Norman J. Haughey.
Cell Death & Disease
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Full TextTransplantation of Cardiac Mesenchymal Stem Cell-Derived Exosomes Promotes Repair in Ischemic Myocardium
Chengwei Ju, Yan Shen, Gengshan Ma, Yutao Liu, Jingwen Cai, Il-man Kim, Neal L. Weintraub, Naifeng Liu, Yaoliang Tang
Journal of Cardiovascular Translational Research
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
Angiogenesiscardiac mesenchymal stem cellscardiac repair therapycardiomyocyte proliferationCMSCexosomesinfarct treatmentischemic myocardiummyocardial infarction
Full TextUrinary Exosomes and Exosomal CCL2 mRNA as Biomarkers of Active Histologic Injury in IgA Nephropathy
Ye Feng, Lin-Li Lv, Wei-Jun Wu, Zuo-Lin Li, JunChen, Hai-Feng Ni et al.
The American Journal of Pathology
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
AlixAquaproin 2CD63exosome markersexosomesIgA nephropathyIgANIgAN diagnosisnephrinnon-invasive biomarkersrenal cell markersrenal disease diagnosisrenal injuryrenal pathologyurinary exosomes
Full TextVery long-chain ceramides are increased in serum exosomes with aging in both human subjects an non-human primates
P Krishnan, A Khayrullin, L Martinez, B Mendhe, S Fulzele, Y Liu, MD, J A Mattison, M W Hamrick
Innovation in Aging
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesicles
AgingApoptosiscardiovascular healthcell senescencememory impairmentserum derived exosomesvery long-chain ceramides
Full TextA Comparative Study of Serum Exosome Isolation Using Differential Ultracentrifugation and Three Commercial Reagents
Inas Helwa, Jingwen Cai, Michelle D. Drewry, Arthur Zimmerman, Michael B. Dinkins, Mariam Lotfy Khaled, Mutsa Seremwe, W. Michael Dismuke, Erhard Bieberich, W. Daniel Stamer, Mark W. Hamrick, Yutao Liu
PLOS ONE
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesZeta Potential
CD63CD9differential ultracentrifugationexosomal RNAhigh-density lipoproteinshsa-miR-16hsa-miR-451human serumpolyethylene glycolTEM
Full TextAn In Vitro Potency Assay for Monitoring the Immunomodulatory Potential of Stromal Cell-Derived Extracellular Vesicles
Karin Pachler, Nina Ketterl, Alexandre Desgeorges, Zsuzsanna A. Dunai, Sandra Laner-Plamberger, Doris Streif, Dirk Strunk, Eva Rohde and Mario Gimona
International Journal of Molecular Sciences
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
exosomesextracellular vesiclesimmune modulationmesenchymalT-cell proliferation
Full TextAtg5 Disassociates the V1V0-ATPase to Promote Exosome Production and Tumor Metastasis Independent of Canonical Macroautophagy
Huishan Guo, Maneka Chitiprolu, Luc Roncevic, Charlotte Javalet, Fiona J. Hemming, My Tran Trung, Lingrui Meng, Elyse Latreille, Christiano Tanese de Souza, Danielle McCulloch, R. Mitchell Baldwin, Rebecca Auer, Jocelyn Côté, Ryan Charles Russell, Rémy Sadoul, Derrick Gibbings
Developmental Cell
2017
ApplicationsCancerConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
AcidificationAtg5Autophagycancerendosomeexosomesextracellular vesiclesLC3multivesicular bodytumorV1V0-ATPase
Full TextBreast-cancer extracellular vesicles induce platelet activation and aggregation by tissue factor-independent and -dependent mechanisms
FG Gomes, V Sandim, VH Almeida, AMR Rondon.
Thrombosis Research
2017
ApplicationsBreast CancerCancerConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
AggregationBreast Cancer Derived EVscancer-associated thrombosisMCF-7 EVsMDA-MB-231 EVsplasma coagulationplatelet activationthrombin generationTissue factor
Full TextEscherichia coli outer membrane vesicles can contribute to sepsis induced cardiac dysfunction
Kristina Svennerholm, Kyong-Su Park, Johannes Wikström, Cecilia Lässer, Rossella Crescitelli, Ganesh V. Shelke, Su Chul Jang, Shintaro Suzuki, Elga Bandeira, Charlotta S. Olofsson & Jan Lötvall.
Scientific Reports
2017
ApplicationsConcentration MeasurementExtracellular VesiclesOuter Membrane VesiclesSize MeasurementVesicles
cardiomyocytescytokineEscherichia coliinflammationLPSOMVouter membrane vesiclesreactive oxygen speciessepsis induced cardiac dysfunctionTroponin T
Full TextExosome production and its regulation of EGFR during wound healing in renal tubular cells
Zhou X, Zhang W, Yao Q, Zhang H, Dong G, Zhang M, Liu Y, Chen JK, Dong Z.
American journal of physiology. Renal physiology
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
CD63EGFRexosomesGW4869kidneymanumycin ATEMtransfer of genetic information between cellsTsg101wound healing
Full TextExtracellular vesicles from KSHV-infected endothelial cells activate the complement system
JL Kang, J Park, SJ Gao, MS Lee.
Oncotarget
2017
ApplicationsConcentration MeasurementExtracellular VesiclesMicroparticlesSize MeasurementTechnical NanoparticlesVesiclesViruses
Alternative Complement PathwayCD63CD81complement systemendothelial cellsextracellular vesiclesherpesvirusHSP70innate immunityKSHVNF-κB pathwayproteomics analysis
Full TextGender-specific differential expression of exosomal miRNA in synovial fluid of patients with osteoarthritis
Kolhe R, Hunter M, Liu S, Jadeja RN, Pundkar C, Mondal AK, Mendhe B, Drewry M, Rojiani MV, Liu Y, Isales CM, Guldberg RE, Hamrick MW, Fulzele S
Scientific Reports - Nature
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
Full TextHIF-1-mediated production of exosomes during hypoxia is protective in renal tubular cells
Wei Zhang, Xiangjun Zhou, Qisheng Yao, Yutao Liu, Hao Zhang, Zheng Dong
American journal of physiology. Renal physiology
2017
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Full TextHigh glucose increases the formation and pro-oxidative activity of endothelial microparticles
Burger, D., Turner, M., Xiao, F. et al.
Diabetologia
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
CD81CD9GM130mesenchymal stem cellsprogenitor cellsstromal cellsTsg101
Full TextIn Vivo Neuroimaging of Exosomes Using Gold Nanoparticles
O Betzer, N Perets, A Angel, M Motiei, T Sadan et al.
ACS Nano
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
BiodistributionBrain Ischemiacomputed tomographyexosome labelling techniqueexosome traffickingexosome-based treatmentexosomesglucose coatingGLUT-1gold nanoparticlesNeuroimaging
Full TextIncreased miR-124-3p in microglial exosomes following traumatic brain injury inhibits neuronal inflammation and contributes to neurite outgrowth via their transfer into neurons
S Huang, X Ge, J Yu, Z Han, Z Yin, Y Li, F Chen et al.
The FASEB Journal
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
Brain Derived Exosomesextracellular vesiclesglia cellsmiRNAmTOR signalingneuronal exosome transferneuronal inflammationPDE4Bpolarizationtraumatic brain injury
Full TextLarger size extracellular vesicle as early biomarker in patients with metastatic pancreatic cancer
Dong Uk Kim, Francis A San Lucas, Matthew Cagley, Bret Stephens, Feven C Mulu, Jonathan Castillo, Vincent Bernard, Gauri R. Varadhachary, Matthew H. G. Katz, Hector A Alvarez, Anirban Maitra;
Journal of Clinical Oncology
2017
ApplicationsCancerConcentration MeasurementExtracellular VesiclesPancreatic CancerSize MeasurementVesicles
Bioactive MoleculesBiomarkerLiquid biopsymetastatispancreatic cancerPDACpredictorstumor progression
Full TextMagnetic nanoparticle-enhanced surface plasmon resonance biosensor for extracellular vesicle analysis
A Reiner, NG Ferrer, P Venugopalan, RC Lai, S Lim.
Analyst
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesZeta Potential
Full TextMesenchymal Stem/Stromal Cell-Derived Extracellular Vesicles and Their Potential as Novel Immunomodulatory Therapeutic Agents
Verena Börger, Michel Bremer, Rita Ferrer-Tur, Lena Gockeln, Oumaima Stambouli, Amina Becic and Bernd Giebel
International Journal of Molecular Sciences
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
AlixAngiogenesisAnti-inflammatory CapabilitiesApoptosisCD63CD81CD9cell proliferationcell therapycytokinesexosomesextracellular vesiclesfibrosisimmunomodulationmesenchymal stem/stromal cellsmicrovesiclesneurogenesisoxidative stressTsg101
Full TextMethods for the physical characterization and quantification of extracellular vesicles in biological samples
Déborah L.M. Ruperta, Virginia Claudio, Cecilia Lässer, Marta Bally
Biochimica et Biophysica Acta (BBA) - General Subjects
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesZeta Potential
Analytical MethodsBuoyant DensityCharacterizationexosomesextracellular vesiclesoptical propertiesPhysical propertiesQuantificationsizestructurevesicle subpopulations
Full TextMicroRNA-183-5p Increases with Age in Bone-Derived Extracellular Vesicles, Suppresses Bone Marrow Stromal (Stem) Cell Proliferation, and Induces Stem Cell Senescence
Davis C, Dukes A, Drewry M, Helwa I, Johnson M, Isales CM, Hill WD, Liu Y, Shi X, Fulzele S, Hamrick MW
Tissue engineering
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
Full TextMining Extracellular Vesicles for Clinically Relevant Noninvasive Diagnostic Biomarkers in Cancer.
Muhammad Nawaz, Farah Fatima and Jeremy A. Squire.
Novel Implications of Exosomes in Diagnosis and Treatment of Cancer and Infectious Diseases
2017
BiomarkersCancerExtracellular VesiclesVesicles
Analytical TechnologiesEV isolationexosomesexRNAextracellular vesiclesgenetic profilingLiquid biopsymetastasismiRNAnoninvasive diagnosis
Full TextMouse serum exosomal proteomic signature in response to asbestos exposure
P Munson, YW Lam, M MacPherson, S Beuschel, A Shukla.
Journal of Cellular Biochemistry
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
AsbestosCD81CD9exosome cargoexosomesmesotheliomaoropharyngeal aspirationproteomic analysisproteomics
Full TextNeutral Sphingomyelinase-2 Deficiency Ameliorates Alzheimer’s Disease Pathology and Improves Cognition in the 5XFAD Mouse
Michael B Dinkins, John Enasko, Caterina Hernandez, Guanghu Wang, Jina Kong, Inas Helwa, Yutao Liu, Alvin V Terry Jr, Erhard Bieberich
The Journal of neuroscience: the official Journal of the Society of Neuroscience
2017
ApplicationsExtracellular VesiclesSize MeasurementVesicles
Full TextNovel Therapy for Glioblastoma Multiforme by Restoring LRRC4 in Tumor Cells: LRRC4 Inhibits Tumor-Infitrating Regulatory T Cells by Cytokine and Programmed Cell Death 1-Containing Exosomes
Peiyao Li, Jianbo Feng, Yang Liu, Qiang Liu, Li Fan, Qing Liu, Xiaoling She, Changhong Liu, Tao Liu, Chunhua Zhao, Wei Wang, Guiyuan Li and Minghua Wu.
Frontiers in Immunology
2017
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize Measurement
CCR4CD63CD81CD9exosomeGBM immune microenvironmentGlioblastomaHSP70LRRC4methylation-mediated LRRC4 inactivationmiRNA expressionprogrammed cell death 1regulatory T cellstumor-infiltrating lymphocytes
Full TextPediatric brain tumor cells release exosomes with a miRNA repertoire that differs from exosomes secreted by normal cells
Ágota Tűzesi, Teresia Kling, Anna Wenger, Taral R. Lunavat, Su Chul Jang, Bertil Rydenhag, Jan Lötvall, Steven M. Pollard, Anna Danielsson, Helena Carén.
Oncotarget
2017
ApplicationsCancerConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
Braincancer stem cellsexosomesgene expressiongliomaGlioma stem cellmicroRNAmRNA expressionneural stem cellpediatric
Full TextPrions on the run: How extracellular vesicles serve as delivery vehicles for self-templating protein aggregates
Shu Liu, André Hossinger, Sarah Göbbels & Ina M. Vorberg.
Prion
2017
ApplicationsConcentration MeasurementExtracellular VesiclesPrion ProteinsSize MeasurementVesicles
2a ModeldisseminationexosomeHEK NM-HAintercellular transmissionneurodegenerative diseasesspreadingSup35 NM priontransmissible spongiform encephalopathiesyeast prions domain
Full TextSingle-Particle Discrimination of Retroviruses from Extracellular Vesicles by Nanoscale Flow Cytometry
Vera A. Tang, Tyler M. Renner, Anna K. Fritzsche, Dylan Burger & Marc-André Langlois.
Scientific Reports - Nature
2017
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesiclesVirusesZeta Potential
eGFPextracellular vesiclesHIVMoloney murine leukemia virusnanoscale flow cytometryRetrovirusSingle particle discriminationVirus-containing supernatant
Full TextThe Effects of High Glucose Exposure on Endothelial Microparticles
Maddison Turner
Cellular and Molecular Medicine, Faculty of Medicine, University of Ottawa
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesZeta Potential
diabeteselectronegativityendothelial damageEndothelial MicroparticlesGlucosehyperglycemic environmentoxidative stressreactive oxygen speciestherapeuticvascular injury
Full TextThe release and trans-synaptic transmission of Tau via exosomes
Wang, Y., Balaji, V., Kaniyappan, S. et al.
Molecular Neurodegeneration
2017
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
AlixAlzheimer Diseaseconditioned mediumexosomal markersFlotillinGFP-TauN2a cellsneuron-to-neuron transmissionTau protein aggregatestrans-synaptic Tau transmission
Full TextAssessment of urinary microparticles in normotensive patients with type 1 diabetes
Yuliya Lytvyn, Fengxia Xiao, Christopher R. J. Kennedy, Bruce A. Perkins, Heather N. Reich, James W. Scholey, David Z. Cherney, Dylan Burger.
Diabetologia
2016
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
AlbuminuriaBiomarkerdiabetesextracellular vesicleHyperglycaemiaMicroparticleMicrovesiclenephrinPodocyterenal injuryurinary vesicles
Full TextDetailed Analysis of Protein Topology of Extracellular Vesicles – Evidence of Unconventional Membrane Protein Orientation
Aleksander Cvjetkovic, Su Chul Jang, Barbora Konečná, Johanna L. Höög, Carina Sihlbom, Cecilia Lässer & Jan Lötvall
Scientific Reports - Nature
2016
ApplicationsConcentration MeasurementExtracellular VesiclesVesicles
CD81EV cargo proteomeHMC-1 mast cell linelipid anchored proteinsMembrane Protein OrientationProtein TopologyProteinase K treatmentsurface-accessible EV proteometransmembrane proteinsTsg101
Full TextHIV-Nef and ADAM17-Containing Plasma Extracellular Vesicles Induce and Correlate with Immune Pathogenesis in Chronic HIV Infection
Jung-Hyun Lee, Stephan Schierer, Katja Blume, Jochen Dindorf, Sebastian Wittki, Wei Xiang, et al.
eBioMedicine
2016
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
ADAM17ARTCD4Chronic HIV infectionextracellular vesiclesHIVHIV reservoirNefT-cellsVpu
Full TextHorizontal Transmission of Cytosolic Sup35 Prions by Extracellular Vesicles
Shu Liu, André Hossinger, Julia P Hofmann, Philip Denner, Ina M Vorberg
mBio
2016
ApplicationsConcentration MeasurementExtracellular VesiclesPrion ProteinsSize MeasurementVesicles
Full TextHuman mast cells release extracellular vesicle-associated DNA
Ganesh V Shelke, Su Chul Jang, Yanan Yin, Cecilia Lässer, Jan Lötv
matters
2016
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesZeta Potential
Aggregation of EVscargocell-free DNAEV fractionEV-associated DNAextracellular DNAmesenchymal stem cellsmiRNAmRNAnegative charge of the vesiclesretrotransposons
Full TextIsolation and characterization of vesicular and non-vesicular microRNAs circulating in sera of partially hepatectomized rats
Mirco Castoldi, Claus Kordes, Iris Sawitza & Dieter Häussinger
Scientific Reports - Nature
2016
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
Full TextMechanism of Fibronectin Binding to Human Trabecular Meshwork Exosomes and Its Modulation by Dexamethasone
Dismuke WM, Klingeborn M, Stamer WD.
PLOS ONE
2016
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesicles
corticosteroidsdexamethasonedipeptidyl peptidase activityFibronectinGlaucomaocular hypertensionTrabecular Meshwork Exosomes
Full TextMSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease
Kordelas, L., Rebmann, V., Ludwig, A.-K., Radtke, S., Ruesing, Jl, Doeppner, T.R., Epple, M., Horn, P.A., Beelen, D.W., and Giebel, B.
Leukemia
2014
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
CD81conditioned mediaEV fractionexosomal markerextracellular vesiclesgraft-versus-host-diseaseimmune-modulating activitiesmicrovesiclesMSC-derived exosomesmultivesicular bodiesTsg101
Full TextIn vivo identification of an HLA?G complex as ubiquitinated protein circulating in exosomes
Estibaliz Alegre, Vera Rebmann, Joel Lemaoult, Carmen Rodriguez, Peter A Horn, Angel Díaz-Lagares, José I Echeveste, Alvaro González
European Journal of Immunology
2013
ApplicationsCancerConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
CD63circulating HLA-G complexesexosomeshuman leukocyte antigen-G (HLA-G)markermicrovesiclesNK cellstumor spreadingUbiquitin
Full TextTumor and Endothelial Cell-Derived Microvesicles Carry Distinct CEACAMs and Influence T-Cell Behavior
Harrison T. Muturi, Janine D. Dreesen, Elena Nilewski, Holger Jastrow, Bernd Giebel, Suleyman Ergun, Bernhard B. Singer
PLOS ONE
2013
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesicles
Angiogenesiscarcinoembryonic antigen related cell adhesion molecule (CEACAM)CEACAM-familyendothelial cellsmetastasismicrovesiclesmultivesicular bodiestumor cellstumor progression
Full TextPublications related to research on viruses
Hydrodynamic characterization of a vesicular stomatitis virus‑based oncolytic virus using analytical ultracentrifugation
Wawra, Simon; Kessler, Sophia; Egel, Arina; Solzin, Johannes; Burkert, Oliver; Hochdorfer, Daniel
European Biophysics Journal
2023
ApplicationsConcentration MeasurementSize MeasurementViruses
Analytical ultracentrifugationnanoparticle tracking analysisOncolytic virusVesicular stomatitis virus
Full TextSingle-Pass Tangential Flow Filtration (SPTFF) of Nanoparticles: Achieving Sustainable Operation with Dilute Colloidal Suspensions for Gene Therapy Applications
Chaubal, Akshay S.; Zydney, Andrew L.
Membranes 13 (4)
2023
ApplicationsIsolation TechniquesMicroparticlesSize MeasurementTechnical NanoparticlesViruses
concentration polarizationcontinuous processingcritical fluxgenegene therapylentivirusnanoparticlesSPTFFtherapyultrafiltrationviral vectors
Full TextDevelopment of surface engineered antigenic exosomes as vaccines for respiratory syncytial virus
Suyeon Hong, Shaobo Ruan, Zachary Greenberg, Mei He, and Jodi L. McGill
Scientific Reports
2021
ExosomesExtracellular VesiclesVaccinesVesiclesVirusesZeta Potential
dendritic cell exoxomesMHCRespiratory syncytial virus
Full TextSalivary extracellular vesicles inhibit Zika virus but not SARS-CoV-2 infection
Carina Conzelmann, Rüdiger Groß, Min Zou, Franziska Krüger, André Görgens, Manuela O Gustafsson, Samir El Andaloussi, Jan Münch & Janis A. Müller
Journal of Extracellular Vesicles
2020
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesViruses
Corona virusEV isolationEV purificationsaEVssalivary EVsSARS-CoV-2Zika virus
Full TextChanges of microRNAs expression profiles from red swamp crayfish (Procambarus clarkia) hemolymph exosomes in response to WSSV infection
HuiYang, XixiLi, Jiaojun Ji, ChunyouYuan, Xiaojian Gao, Yingying Zhang, Cheng Lu, Fenggang Li, Xiaojun Zhang
Fish & Shellfish Immunology
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesViruses
deep sequencingexosomeexosomesfishimmunologymicroRNAmiRNAP. clarkiaprocambarus clarkiashellfishWSSVWSSV virus
Full TextExosomes Released from Rabies Virus-Infected Cells May be Involved in the Infection Process
Jingyu Wang, Fan Wu. Chuntian Liu, Wenwen Dai, Yawei Teng, Weiheng Su, Wei Kong, Feng Gao et al.
Virologica Sinica
2019
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesViruses
exosome isolationexosomesrabiesrabies infectionrabies virus
Full TextAntiretroviral Drugs Alter the Content of Extracellular Vesicles from HIV-1-Infected Cells
C DeMarino, ML Pleet, M Cowen, RA Barclay et al.
Scientific Reports
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesViruses
cART-altered EV contentCD63CD81CD9DemeclocyclineDoxycyclineextracellular vesiclesFDA-approved drugsHIV-1MethacyclineMinocyclinenon-coding viral RNAOxycyclineOxytetracyclineSCRT pathwayTAR RNtetracyclineVPS4
Full TextEbola Virus VP40 Modulates Cell Cycle and Biogenesis of Extracellular Vesicles
Michelle L Pleet, James Erickson, Catherine DeMarino, Robert A Barclay, Maria Cowen, Benjamin Lepene, Janie Liang, Jens H Kuhn, Laura Prugar, Spencer W Stonier, John M Dye, Weidong Zhou, Lance A Liotta, M Javad Aman, Fatah Kashanchi
The Journal of Infectuos Diseases
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementTechnical NanoparticlesVesiclesViruses
Apoptosiscell cycleebola treatmentEbola virusEBOVEBOV pathogenesiEBOV treatmentEV biogenesisEVsextracellular vesiclesmonocyte apoptosispoptosis; cell cycleT cell apoptosisVP40
Full TextExosome markers associated with immune activation and oxidative stress in HIV patients on antiretroviral therapy
S Chettimada, DR Lorenz, V Misra, ST Dillon et al.
Scientific Reports
2018
ApplicationsConcentration MeasurementExtracellular VesiclesSize MeasurementVesiclesViruses
CD63CD81CD9CRPD14exosome cargoHIVHLA-AHLA-Bimmunological markersimmunomodulationoxidative stressplasma exosome
Full TextNef Secretion into Extracellular Vesicles or Exosomes Is Conserved across Human and Simian Immunodeficiency Viruses
Ryan P. McNamara, Lindsey M. Costantini, T. Alix Myers, Blake Schouest, Nicholas J. Maness, Jack D. Griffith, Blossom A. Damania, Andrew G. MacLean, Dirk P. Dittmer.
mBio
2018
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesiclesViruses
AIDSCD4CD63CD81exosomesextracellular vesiclesFlotillinHIVlentivirusesmicrovesiclesNefSIV
Full TextExtracellular vesicles from KSHV-infected endothelial cells activate the complement system
JL Kang, J Park, SJ Gao, MS Lee.
Oncotarget
2017
ApplicationsConcentration MeasurementExtracellular VesiclesMicroparticlesSize MeasurementTechnical NanoparticlesVesiclesViruses
Alternative Complement PathwayCD63CD81complement systemendothelial cellsextracellular vesiclesherpesvirusHSP70innate immunityKSHVNF-κB pathwayproteomics analysis
Full TextSingle-Particle Discrimination of Retroviruses from Extracellular Vesicles by Nanoscale Flow Cytometry
Vera A. Tang, Tyler M. Renner, Anna K. Fritzsche, Dylan Burger & Marc-André Langlois.
Scientific Reports - Nature
2017
ApplicationsConcentration MeasurementExosomesExtracellular VesiclesSize MeasurementVesiclesVirusesZeta Potential
eGFPextracellular vesiclesHIVMoloney murine leukemia virusnanoscale flow cytometryRetrovirusSingle particle discriminationVirus-containing supernatant
Full Text