Nanoparticle Tracking Analysis Concentration Zeta Potential Fluorescence Size Colocalization
Nanoparticle Tracking Analysis
Nano-particle
Tracking
Analysis
Nanoparticle Tracking Analysis (NTA) is a technique to characterize nanoparticles in suspension in the size range of 10 – 2000 nm. A microscopic setup allows real-time visualization of particles below the diffraction limit of conventional microscopes, in both scatter and fluorescence mode. A broad range of applications from nano-sized metallic particles to polymers and biological nanoparticles such as viruses or extracellular vesicles makes NTA a versatile method for sizing – independent of the refractive index of the particle material.
Application Range
Traditionally, NTA has been used for concentration and size measurement of nanoparticles dispersed in a medium. In addition, Particle Metrix ZetaView® provides zeta potential for surface charge and characterization of fluorescently labeled bionanoparticles. This enables NTA to perform multiparameter measurements for all types of particles on the same sample, saving operator time and sample quantity. Applications of NTA range from quantifying the size, concentration or zeta potential of inorganic particles such as silicon dioxide, polymers, calcites, phosphates, barium sulfate to carbon nanotubes or nanobubbles. Furthermore, all types of bionanoparticles (BNP) such as protein aggregates, extracellular particles (such as exosomes or EVs), viruses or liposomes can be analyzed and by fluorescence labeling without any additives.
Metallic Nanoparticles
Metallic Nanoparticles
Extracellular Vesicles
Extracellular Vesicles
Inorganic Particles
Inorganic Particles
Viruses
Viruses
Special "Particles"
Special "Particles"
Bionanoparticles
Bionanoparticles
Magnetic Particles
Magnetic Particles
Fluorescent Standard Particles
Fluorescent Standard Particles
History
In 1828, Brown discovered the random walk behavior of pollen [1]. However, it took about 80 years to lay the theoretical foundation for the method named after him: Brownian motion analysis [2, 3]. In the early 1990s, there were the first publications on the use of computers for the automated analysis of images [4]. Commercial implementation of this technique required the availability of fast computer systems that allowed computationally intensive video analysis in reasonable time frames.
Today, NTA is an established technique for the characterization of nanoparticles; a detailed procedure for the practical procedure for NTA measurement is given in the ASTM standard for NTA measurements [5]. For the characterization of bionanoparticles, Nanoparticle Tracking Analysis is part of the “Minimal information for studies of EVs” (MISEV) [6-8].
Physical Principle
The particles in the sample are visualized by illumination with a laser beam. The scattered light from the particles is recorded with a light-sensitive CMOS camera chip placed at a 90° angle to the illumination plane. The setup, also known as ultra microscopy, [9] allows detection and recording of each individual particle below the size limit of typical microscopes. The size of each particle is calculated by Brownian motion analysis of the individual tracks, allowing simultaneous determination of size and concentration.
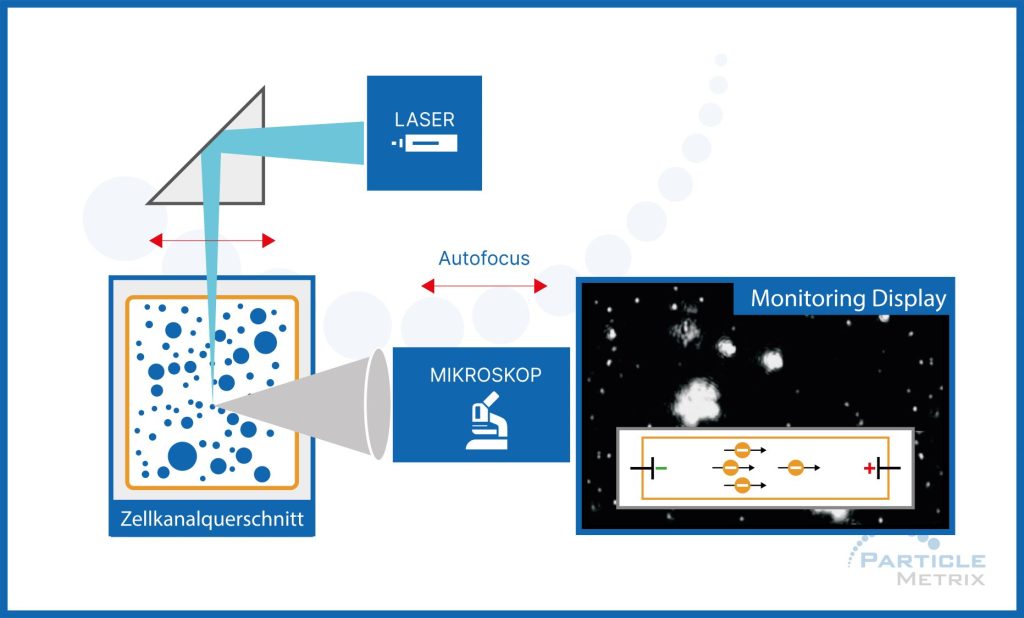
A brief introduction to the physical principle underlying NTA is as follows:
When small particles are dispersed in a liquid, the particles move randomly in all directions. The liquid is also called the continuous phase and is typically water, PBS, or physiological buffer, but can also be an organic solvent such as ethanol or cyclohexane. The phenomenon of random motion is termed diffusion and is expressed by the diffusion coefficient D . More specifically, the undirected migration of given particles is caused by energy transfer from surrounding water molecules to the particle. The theory for determining the diffusion coefficient was developed by the famous physicist Albert Einstein [2]. In the absence of a concentration gradient within the dispersion and with long-term observation, the distance traveled by small particle in any direction should cancel out over time, so that the total motion is nearly zero. However, during certain time intervals, diffusing particles move within certain volume elements. In NTA the time t between two observations is quite short (~30 ms). The motion of particles per time interval is recorded and quantified as the mean square displacement . Depending on the number of dimensions (one, two or all three dimensions) the observed mean square displacement, the diffusion coefficient, can be calculated as follows:
Using the Stokes-Einstein relationship the particle diameter d can be calculated as function of the diffusion coefficient D at a temperature T and a viscosity η of the liquid ( k_B Boltzmann constant),
In Nanoparticle Tracking Analysis, the particle fluctuation of a single particle is recorded in two dimensions. After combining the Stokes-Einstein relationship and the two-dimensional mean square displacement, the equation for the particle diameter d can be solved with:
Size Range
The lower limit of the working range i.e. the smallest detectable particle size, depends on the scattering intensity of the particle, the efficiency of the magnification optics and the sensitivity of the camera. Metallic nanoparticles are strong scatterers due to their comparatively large refractive index, allowing detection down to sizes of ~10 nm. Biological nanoparticles such as EVs have refractive indices of around 1.37-1.45 resulting in a limit of detection of 30-50 nm for Nanoparticle Tracking Analysis [10].
Nanoparticle Tracking Analysis
is a Sensitive Technique
Nanoparticle Tracking Analysis achieves low concentrations of ~105 particles per mL, and can even be used for detecting certain tracing impurities. Typically, impurities can originate in the form of particles from the buffer (distilled water or buffering agents) and are unknowingly generated during sample preparation as precipitates of phosphates, carbonates or silicates, or as bubbles. When used at high concentrations near their critical micellar concentration (CMC), stabilizing agents such as surfactants are likely to show foaming and generate nanobubbles. The high sensitivity of NTA is ensured by filtration of buffers, degassing and verified by blank controls.
To understand the underlying processes in your sample, zeta potential measurement and fluorescence detection are integrated for comprising particle characterization. Subpopulation detection based on multivariate statistics takes resolution of NTA to the next level.
Nanoparticle Tracking Analysis
is a Multi-Parameter Technique
Zeta Potential
Zeta Potential
Subpopulations
Subpopulations
Concentration
Concentration
Fluorescence
Fluorescence Detection
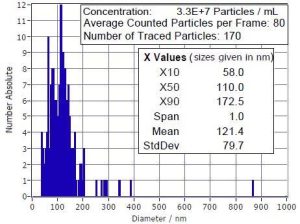
Size
Particle Size Distribution
Colocalization
Colocalization
Summary
Nanoparticle Tracking Analysis is a particle characterization technique for virtually all types of nanoparticles suspended in media. The size, concentration, zeta potential and fluorescence are measured on the same sample.
The highlights of this technique are:
- Colocalization
- Characterization of nanoparticles in solution
- Measurement at comparatively low concentration levels (down to 105 particles per cm3)
- Single particle analysis resulting in high-resolution distributions
- Specificity with fluorescent detection
NTA is a fast single particle analysis tool for visualization and quantification of subpopulations, size, fluorescence, surface charge (zeta potential) and concentration.
References
[1] Brown, Robert (1828). “A brief account of microscopical observations made in the months of June, July and August, 1827, on the particles contained in the pollen of plants; and on the general existence of active molecules in organic and inorganic bodies” (PDF). Philosophical Magazine. 4 (21): 161–173. doi:10.1080/14786442808674769
[2] Einstein, A., Annalen der Physik, 19 (1906), Seiten 371-381, Zur Theorie der Brownschen Bewegung
[3] Smoluchowski, von, M., Annalen der Physik, 14, Seiten 756-780, 1906, Zur kinetischen Theorie der Brownschen Molekularbewegung und der Suspensionen https://doi.org/10.1002/andp.19063261405
[4] Qian, H., Sheets, M. P., Elson, E. L., Single particle tracking, Biophys J. 1991 Oct; 60(4): 910–921. doi: 10.1016/S0006-3495(91)82125-7
[5] ASTM E2834-12, Standard Guide for Measurement of Particle Size Distribution of Nanomaterials in Suspension by Nanoparticle Tracking Analysis (NTA), ASTM International, West Conshohocken, PA, 2012, www.astm.org
[6] Updating the MISEV minimal requirements for extracellular vesicle studies: building bridges to reproducibility https://doi.org/10.1080/20013078.2017.1396823
[7] Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines https://doi.org/10.1080/20013078.2018.1535750
[8] Giebel B, Helmbrecht C. Methods to Analyze EVs. Methods Mol Biol. 2017;1545:1-20. doi: 10.1007/978-1-4939-6728-5_1. PMID: 27943203.
[9] Zsigmondy, R., Colloids and the Ultra Microscope, J. Am. Chem. Soc., 1909, 31 (8), pp 951–952. doi: 10.1021/ja01938a017
[10] Bohren CF, Huffman DR. Absorption and scattering by a sphere. In: Bohren CF, Huffman DR, editors. Absorption and scattering of light by small particles. Weinheim: Wiley-VCH Verlag GmbH; 2007. pp. 83–129.
Read More

High Throughput ZetaView® System vs High Cost of Other Methods
In addition to using Nano Particle Tracking Analysis (NTA) to measurement the size distribution and concentration of EV samples, both Microfluidic Resistive Pulse Sensing (MRPS) and the Single Particle Interference Reflectance Image Sensor (SP-IRIS) methods have been widely used as an alternative means of characterizing EVs. In this note, we relay two specific cases for using the ZetaView® NTA system to achieve relative “high-throughput” analysis of many EV samples, along estimations of throughput for MRPS & SPIRIS methods for an equivalent number of samples; further, we establish realistic estimates for the high cost of ownership for operating MRPS & SP-IRIS systems as a result of the cost of consumables as well as the substantially greater amount of time spent to run the same number of samples.
Avoiding Artifacts in EV samples:
Part 1 – the Use of Tween®
Along with generally accepted methods are some “tricks of the trade” such as the addition of Tween® or BSA; however, some of those additions are problematic.
Principles of Colocalization-NTA
Platinum Presenter Talk at ISEV 2022 in Lyon Dr. Clemens Helmbrecht NTA is a multi-parameter technology Nanoparticle Tracking Analysis (NTA) is well known for measuring
Discover Applications
EXTRACELLULAR VESICLES
Research on Exacellular Vesicles is thriving. Nanoparticle Tracking Analysis plays an important role in this development due to in vitro measurement of multiple physical parameters such as size, concentration, surface charge, and phenotype characteristics
Viruses
Nanoparticle Tracking Analysis can be used to quantify total concentration of virions, analyse the amount of successfully loaded virions as well as virus aggregatiion.