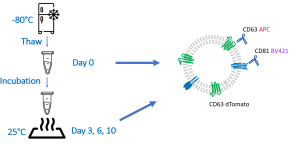
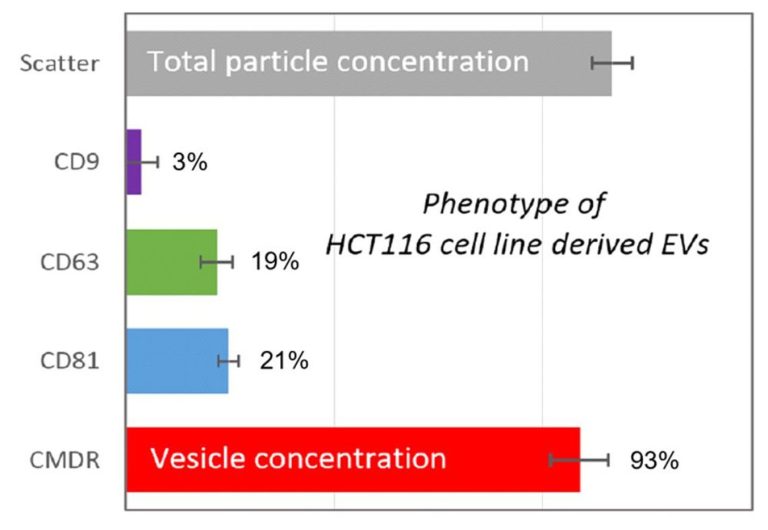
Andreu Z. and Yáñez-Mó M. Tetraspanins in Extracellular Vesicle Formation and Function. Front Immunol. 2014; 5: 442
Bachurski D., Schuldner M., Nguyen P-H., Malz A., Reiners K.S., Grenzi P.C., Babatz F., Schauss A.C., Hansen H.P., Hallek M. and Pogge von Strandmann E. Extracellular vesicle measurements with nanoparticle tracking analysis – An accuracy and repeatability comparison between NanoSight NS300 and ZetaView. J. Extracell Vesicles. 2019: 8(1): 1596016.
Baixauli F., Lopez-Otin C., Mittelbrunn M. Exosomes and autophagy: coordinated mechanisms for the maintenance of cellular fitness. Front Immunol. 2014; 5:403.
Becker A., Thakur B., Weiss J. M., Kim H. S., Peinado H., Lyden D. Extracellular Vesicles in Cancer: Cell-to-Cell Mediators of Metastasis. Cancer Cell. 2016 Dec 12;30(6):836-848.
Berditchevski F. and Odintsova E. Tetraspanins as Regulators of Protein Trafficking. Traffic 2007; 8:89–96.
Console L., Scalise M., Indiveri C. Exosomes in inflammation and role as biomarkers. Clin Chim Acta. 2019; 488:165-171.
Deatherage B.L. and Cookson B.T. Membrane Vesicle Release in Bacteria, Eukaryotes, and Archaea: a Conserved yet Underappreciated Aspect of Microbial Life. Infect Immun. 2012; 80(6):1948-57.
Desgeorges A., Hollerweger J., Lassacher T., Rohde E., Helmbrecht C., and Gimona M. Differential fluorescence nanoparticle tracking analysis for enumeration of the extracellular vesicle content in mixed particulate solutions. Methods. 2020 Feb 17. pii: S1046-2023 (19)30264-6.
Giebel B. and Helmbrecht C. Methods to Analyze EVs; Methods Mol Biol. 2017; 1545:1-20.
Huang S., Yuan S., Dong M., Su J., Yu C., Shen Y., Xie X., Yu Y., Yu X., Chen S., Zhang S., Pontarotti P. and Xu A. The phylogenetic analysis of tetraspanins projects the evolution of cell-cell interactions from unicellular to multicellular organisms. Genomics. 2005 Dec; 86(6):674-84.
Konoshenko M.Y., Lekchnov E.A., Vlassov A.V., Laktionov P.P. Isolation of Extracellular Vesicles: General Methodologies and Latest Trends. Biomed Res Int. 2018: 8545347.
Mathivanan S., Ji H., Simpson R.J. Exosomes: extracellular organelles important in intercellular communication. J Proteom. 2010;73(10):1907–1920.
Rahbari M, Pecqueux M, Aust D, Stephan H., Tiebel O. Chatzigeorgiou A., Tonn T., Baenke F., Rao V., Ziegler N., Greif H., Lin K., Weitz J., Rahbari N.N. and Kahlert C. Expression of Glypican 3 is an Independent Prognostic Biomarker in Primary Gastro-Esophageal Adenocarcinoma and Corresponding Serum Exosomes. J Clin Med. 2019;8(5):696.
Rajput A, Dominguez San Martin I, Rose R, Beko A., Levea C., Sharratt E., Mazurchuk R., Hoffman R.M., Brattain M.G. and Wang J. Characterization of HCT116 human colon cancer cells in an orthotopic model. J Surg Res. 2008;147(2): 276–281.
Raposo G. and Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200(4):373–383.
Record M., Carayon K., Poirot M., Silvente-Poirot S. Exosomes as new vesicular lipid transporters involved in cell-cell communication and various pathophysiologies. Biochim Biophys Acta. 2014;1841(1):108–120.
Ren R., Sun H., Ma C. , Liu J. and Wang H. Colon cancer cells secrete exosomes to promote self-proliferation by shortening mitosis duration and activation of STAT3 in a hypoxic environment. Cell Biosci. 2019; 9:62
Soo C.Y., Song Y., Zheng Y., Campbell E.C., Riches A.C., Gunn-Moore F. and Powis S.J. Nanoparticle tracking analysis monitors microvesicle and exosome secretion from immune cells. Immunology. 2012, 136:192–197.
Stanly C., Fiume I., Capasso G., Pocsfalvi G. Isolation of Exosome-Like Vesicles from Plants by Ultracentrifugation on Sucrose/Deuterium Oxide (D2O) Density Cushions. Methods Mol Biol. 2016; 1459:259-69.
Vlassov A.V., Magdaleno S., Setterquist R. and Conrad R. Exosomes: Current knowledge of their composition, biological functions, anddiagnostic and therapeutic potentials. Biochimica et Biophysica Acta (BBA). 2012 Jul; 1820(7): 940-948
Weber A., Wehmeyer J.C., Schmidt V., Lichtenberg A. and Akhyari P. Rapid Fluorescence-based Characterization of Single Extracellular Vesicles in Human Blood with Nanoparticle-tracking Analysis. J Vis Exp. 2019 Jan 7;(143)
Yanez-Mo M., Siljander P.R., Andreu Z., Zavec A.B., Borras F.E., Buzas E.I., Buzas K., Casal E., Cappello F., Carvalho J., Colas E., Cordeiro-da Silva A., Fais S., Falcon-Perez J.M., Ghobrial I.M., Giebel B., Gimona M., Graner M., Gursel I., Gursel M., Heegaard N.H., Hendrix A., Kierulf P., Kokubun K., Kosanovic M., Kralj-Iglic V., Kramer-Albers E.M., Laitinen S., Lasser C., Lener T., Ligeti E., Line A., Lipps G., Llorente A., Lotvall J., Mancek-Keber M., Marcilla A., Mittelbrunn M., Nazarenko I., Nolte-’t Hoen E.N., Nyman T.A., O’Driscoll L., Olivan M., Oliveira C., Pallinger E., Del Portillo H.A., Reventos J., Rigau M., Rohde E., Sammar M., Sanchez-Madrid F., Santarem N., Schallmoser K., Ostenfeld M.S., Stoorvogel W., Stukelj R., Van der Grein S.G., Vasconcelos M.H., Wauben M.H., De Wever O. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015; 4:27066.
Zhang Z., Tang C., Zhao L., Xu L., Zhou W., Dong Z., Yang Y., Xiec Q. and Fang X. Aptamer-based fluorescence polarization assay for separation-free exosome quantification. Nanoscale, 2019, 11: 10106-10113