Use of Fluorescence Nanoparticle Tracking Analysis as a tool for quality assessment of EVs
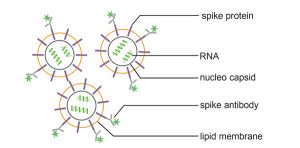
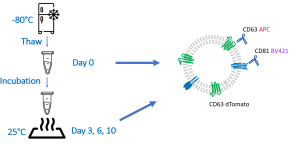
Correct and standardized storage conditions of extracellular vesicles (EVs) are essential prerequisites for the stability of clinical samples and the production of consistent EV batches for therapeutic use.
The loss of EV concentration due to improper storage conditions is associated with a reduced activity and physical integrity.
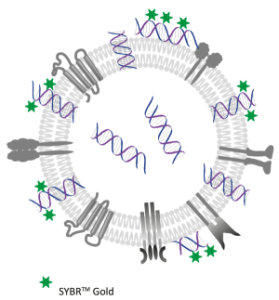
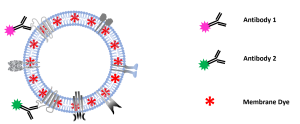
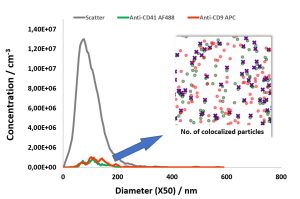
Check out our new app note on the importance of EV storage conditions and how fluorescence nanoparticle tracking analysis can be used as a quality assessment tool.